Dr. Lars Allmendinger
| Dr. Lars Allmendinger Department für Pharmazie - Zentrum für Pharmaforschung - Ludwig-Maximilians-Universität München Butenandtstraße 7 D-81377 München Zi.Nr.: C0.065 Tel.: +49 (0)89 2180 77803 Mail: Lars.Allmendinger@cup.uni-muenchen.de |
 |
Aufgabenbereiche
Analytik
Lehre
Dissertation
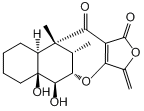
Studien zur Totalsynthese des Antibiotikums Tetrodecamycin
Mitgliedschaften
Deutsche Pharmazeutische Gesellschaft
Gesellschaft Deutscher Chemiker, Fachgruppe Magnetische Resonanzspektroskopie

![]()
Publikationen
33. B. Teng, P. Mandal, L. Allmendinger, C. Douat, Y. Ferrand, I. Huc "Controlling aromatic helix dimerization in water by tuning charge repulsions", Chem. Sci., 2023, Advance Article. (DOI)
32. M. Muhr, H. Liang, L. Allmendinger, R. Bühler, F E. Napoli, D. Ukaj, M. Cokoja, C. Jandl, S. Kahlal, J.-Y. Saillard, C. Gemel, R. A. Fischer "Catalytic Alkyne Semihydrogenation with Polyhydride Ni/Ga Clusters", Angew. Chemie, 2023, (DOI).
31. B. Teng, J. Atcher, L. Allmendinger, C. Douat, Y. Ferrand, I. Huc "Combining local conformational preferences and solvophobic effects in helical aromatic oligoamide foldamers", Org. Biomol. Chem., 2023, 21, 3225-3530 (DOI).
30. F. S. Menke, B. Wicher, L. Allmendinger, V. Maurizot, I. Huc "An abiotic, tetrameric, eight-helix bundle.", Chem. Sci., 2023, 14, 3742-3751. (DOI).
29. M. Währa, L. Allmendinger, G. Höfner, K. T. Wanner, "Benocyclidine (BTCP) as Non-labelled Reporter Ligand for MS Binding Assays for the PCP Ion Channel Binding Site of the Desensitized Torpedo Nicotinic Acetylcholine Receptor (nAChR)", ChemMedChem., 2023, online. (DOI).
28. F. S. Menke, D. Mazzier, B. Wicher, L. Allmendinger, B. Kauffmann, V. Maurizot, I. Huc "Molecular torsion springs: alteration of helix curvature in frustrated tertiary folds.", Org. Biomol. Chem., 2023, 21, 1275-1283. (DOI).
27. D. Bindl, P. K. Mandal, L. Allmendinger, I. Huc "Discrete Stacked Dimers of Aromatic Oligoamide Helices.", Angew. Chemie, 2022, 61, e202116509. (DOI).
26. S. Dengler, P. K. Mandal, L. Allmendinger, C. Douat, I. Huc "Conformational interplay in hybrid peptide-helical aromatic foldamer macrocycles.", Chem. Sci., 2021, 12, 11004. (DOI).
25. T. Ackermann, L. Allmendinger, G. Höfner, K. T. Wanner "MS Binding Assays for Glycine Transporter 2 (GlyT2) Employing Org25543 as Reporter Ligand.", ChemMedChem, 2021, 16(1), 199-215. (DOI).
24. C. A. Karg, P. Wang, F. Kluibenschedl, T. Müller, L. Allmendinger, A. M. Vollmar, S. Moser "Phylloxanthobilins are Abundant Linear Tetrapyrroles from Chlorophyll Breakdown with Activities Against Cancer Cells.", Eur. JOC, 2020. (DOI).
23. D. B. Konrad, G. Savasci, L. Allmendinger, D. Trauner, C. Ochsenfeld, A. M. Ali "Computational Design and Synthesis of a Deeply Red-Shifted and Bistable Azobenzene.", J. Am. Chem. Soc., 2020. (DOI).

22. M. Daerr, L. Allmendinger, G. Höfner, K. T. Wanner "Synthesis and biological evaluation of fluorescent GAT-ligands based on asymmetric substituted BODIPY dyes.", Medicinal Chemistry Research, 2020, 29, 767–782 . (DOI).
21. C. A. Karg, C. M. Schilling, L. Allmendinger, S. Moser, "Isolation, characterization, and antioxidative activity of a dioxobilin-type phylloxanthobilin from savoy cabbage.", Journal of Porphyrins and Phthalocyanines, 2019, 23, 881-888. (DOI).
20. S. Petz, L. Allmendinger, P. Mayer, K. T. Wanner, "Construction of 4-substituted 2-(pyrrolidine-3-yl)acetic acid derivatives as cyclic γ-aminobutyric acid analogues employing intermolecular [2+2]-photocycloaddition as key steps.", Tetrahedron, 2019, 75, 2755-2762. (DOI).
19. P. Neiens, A. De Simone, A. Ramershoven, G. Höfner, L. Allmendinger, K.T. Wanner, "Development and validation of an LC-ESI-MS/MS method for the quantification of D-84, reboxetine, and citalopram for their use in MS Binding Assays addressing the monoamine transporters hDAT, hSERT, and hNET.", Biomedical Chromatography, 2018;32:e4231 (DOI).
18. T. Wein, M. Petrera, L. Allmendinger, G. Höfner, J. Pabel, K. T. Wanner, "Different Binding Modes of Small and Large Binders of GAT1" ChemMedChem 2016, 11, 509–518. (DOI)
17. M. Petrera, T. Wein, L. Allmendinger, M. Sindelar, J. Pabel, G. Höfner, K. T. Wanner, "Development of Highly Potent GAT1 Inhibitors: Synthesis of Nipecotic Acid Derivatives by Suzuki-Miyaura Cross-Coupling Reactions” ChemMedChem 2016, 11, 519–538. (DOI)
16. L. Allmendinger, K. T. Wanner, ”Synthesis of [2H7]indatraline” J. Label Compd. Radiopharm 2014, 57, 721-724. (DOI)
15. S. H. Grimm, L. Allmendinger, G. Höfner, K. T. Wanner, "Enantiopurity Determination of the Enantiomers of the Triple Reuptake Inhibitor Indatraline” Chirality, 2013, 25, 923-933. (DOI)
14. I. Sitka, L. Allmendinger, G. Fülep, G. Höfner, K. T. Wanner, "Synthesis of N-substituted acyclic β-amino acids and their investigation as GABA uptake inhibitors" Eur. J. Med. Chem., 2013, 65, 487-499. (DOI)

13. J. Pabel, M. Faust, C. Prehn, B. Wörlein, L. Allmendinger, G. Höfner, K. T. Wanner, "Development of a (S)-1-{2-[tris(4-methoxyphenyl)methoxy]ethyl}piperidine-3-carboxylic acid [(S)-SNAP-5114] carba analogue inhibitor for murine y-aminobutyric acid transporter type 4" ChemMedChem, 2012, 7, 1245 – 1255. (DOI)
12. C. D. Mayer, L. Allmendinger, F. Bracher, "Synthesis of novel steroid analogues containing nitrile and disulfide moieties via palladium-catalyzed cross-coupling reactions" Tetrahedron, 2012, 6, 1810-1818. (DOI)

11. I. Wetzel, L. Allmendinger, F. Bracher, "Revised Structure of the Alkaloid Drymaritin" J. Nat. Prod., 2009, 72, 1908-1910. (DOI)
10. J. H. Spatz, S. J. Welsch, D.-E. Duhaut, N. Jaeger, T. Boursier, M. Fredrich, L. Allmendinger, G. Ross, J. Kolb, C. Burdack, M. Umkehrer, "Tetramic acid derivatives via Ugi-Dieckmann-reaction" Tetrahedron Lett., 2009, 50, 1705-1707. (DOI)
9. V. Knorr, V. Russ, L. Allmendinger, M. Ogris, E. Wagner, "Acetal Linked Oligoethylenimines for Use As pH-Sensitive Gene Carriers" Bioconjugate Chem., 2008, 19, 1625–1634. (DOI)
8. V. Knorr, L. Allmendinger, G. F. Walker, F. F. Paintner, E. Wagner, "An Acetal-Based PEGylation Reagent for pH-Sensitive Shielding of DNA Polyplexes" Bioconjugate Chem., 2007, 18, 1218-1225. (DOI)
7. F. F. Paintner, L. Allmendinger, G. Bauschke, "A New, General Method for the Highly Regioselective Introduction of Substiutents into the 3-Position of 5-Unsubstituted 4-O-Alkyl Tetronates" Synlett, 2005, 18, 2735-2738. (DOI)
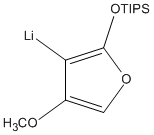
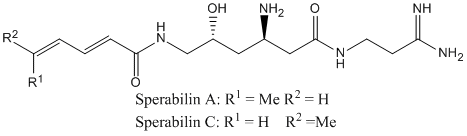
6. L. Allmendinger, G. Bauschke, F. F. Paintner, "Total Synthesis of Sperabilin A and C" Synlett, 2005, 17, 2615-2618. (DOI)
5. F. F. Paintner, L. Allmendinger, G. Bauschke, P. Klemann, "Highly Efficient Approach to Orthogonally Protected (2S,4R)- and (2S,4S)-4-Hydroxyornithine" Org. Lett., 2005, 7, 1423-1426. (DOI)
4. F. F. Paintner, L. Allmendinger, G. Bauschke, C. Berns, P. Heisig, "Synthesis and Antimicrobial Activity of Tetrodecamycin Partial Structures" Bioorg. & Med. Chem., 2003, 11, 2823-2833. (DOI)
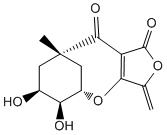
3. F. F. Paintner, L. Allmendinger, G. Bauschke, "Highly Efficient 4-O-Alkylations of Tetronic Acids Involving Oxyphosphonium Intermediates" Synlett, 2003, 1, 83-86. (DOI)
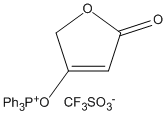
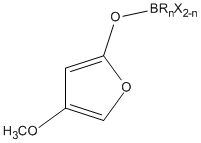
2. F. F. Paintner, L. Allmendinger, G. Bauschke, K. Polborn, "Synthetic Studies toward Tetrodecamycin: An Efficient Approach to the Core Structure of the Antibiotic" Synlett, 2002, 8, 1308-1312. (DOI)
1. F. F. Paintner, L. Allmendinger, G. Bauschke, "Highly Regioselective 3-Hydroxyalkylations of Boron 4-Methoxy-2-furanolates: A New Entry to 5-Unsubstituted and 5-Monosubstituted 3-Acyl-4-O-Methyl Tetronates" Synthesis, 2001, 14, 2113-2118. (DOI)