Research
Biological roles of lanthanides
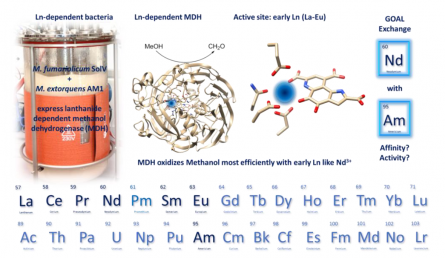
From technology applications in solar cells, mobile phones, batteries and lasers to counterfeiting tags in our Euro currency, lanthanides (Ln) have steadily made their way into our everyday lives. Together with Sc, Y and La, the lanthanides belong to the group of rare earth elements (REE). The term ‘rare’ is clearly misleading as many REEs are abundant, with Ce found in similar concentrations as Cu and Zn in the earth’s crust. What is currently lacking is an understanding and appreciation of the role of REEs in general, but especially the lanthanides (Ln), in biology. Remarkably, in 2014 it was discovered by H. Op den Camp, A. Pol and coworkers (A. Pol et al, Environ. Microbiol., 2014, 16, 255-264) that a methanotrophic microbe (SolV), isolated from volcanic mudpots near Naples, Italy, depends on lanthanides for growth. The respective enzyme responsible for this dependence is a methanol dehydrogenase (MDH). Together with Huub Op den Camp and Arjan Pol we have investigated the first europium containing methanoldehydrogenase and how differnet REE impact bacterial growth and the activity of REE-dependent enzymes.



Our groups aims to gain a deeper understanding for lanthanides in those systems. We are using model complexes, kinetic studies of the enzymes themselves and various spectroscopic techniques to elucidate the mechanism of action of this Ln-dependent MDH.
For an overview on the topic of lanthanides in biology please check out one of our reviews:
L. J. Daumann, Angew. Chem. Int. Ed. 2019, 58, 12795. (In german and englisch available)
B. Jahn and L.J. Daumann, Chem. Unserer Zeit 2018, 52, 150 (In german, open access!)
H. Lumpe and L.J. Daumann, Nachr. Chem. 2018, 66, 945 (In german, free access for GDCh members)
Some book chapters on the topic coming soon!


Half of the team members on this project work with lanthanide dependent bacteria while others work on this topic from the perspective of a coordination chemist
Studies from our lab on Ln-dependent enzymes
First Europium dependent enzyme B. Jahn, A. Pol, H. Lumpe, T. R. M. Barends, A. Dietl, C. Hogendoorn, H. J. M. Op den Camp, L. J. Daumann, ChemBioChem 2018, 19, 1147. (open access, one of the most read, cited articles 2018-2020 in Chembiochem, readers choice)
Impact of the Ln-Contration on Ln-dependent enzymes H. Lumpe, A.Pol, H. J.M. Op den Camp and L. J. Daumann Dalton Trans., 2018,47, 10463-10472 (open access, Back cover)
How to conduct reporducible Ln-MDH assays B.Jahn, N.S.W. Jonasson, H. Hu, H. Singer, A. Pol, N.M. Good, H.J.M. Op den Camp, N.C. Martinez-Gomez and L.J. Daumann, J Biol Inorg Chem 2020, 25, 199. (open access)
Electrochemistry of Ln-MDh with cytochrome P. Kalimuthu, L. J. Daumann, A. Pol, H. J. M. Op den Camp, P. V. Bernhardt, Chem. Eur. J. 2019, 25, 8760. (open access, Cover)
Characterization of the cytochrome partner of Ln-MDH W. Versantvoort, A. Pol, L.J. Daumann, J. Larrabee, A. Strayer, M. Jetten; L. van Niftrik, J. Reimann, H. Op den Camp, BBA - Proteins and Proteomics, 2019, 1867, 6, 595. (open access)
Model studies and studies of redox cofactor PQQ
PQQ interaction with Ln and Ca H. Lumpe and L.J. Daumann, Inorg. Chem. 2019, 58, 13, 8432-8441
Ln-uptake by bacteria
A quick assay to determine Ln-concentration sin bacterial media C. Hogendoorn , P. Roszczenko-Jasińska , N. C. Martinez-Gomez , J. de Graaff , P. Grassl , A. Pol , H. J.M. Op den Camp , L.J. Daumann, Appl. Environ. Microbiol. 2018, 84, e02887-17. (open access)
Biological roles of actinides
One newly established project in the lab deals with the investigation whether actinides (An) can be used instead of Ln in Ln-dependent bacteria and enzymes. Stay tuned!
Mechanistic studies of TET-enzyme model complexes for the elucidation of epigenetically relevant demethylation mechanisms
Here we examine the key steps in the catalytic cycle of AlkBH- and TET-enzymes with the help of spectroscopic techniques and investigation of model complexes that mimic the function of these enzymes. Through a combined approach of rational design of model complexes paired with spectroscopic studies of the enzymatic systems, we hope to unravel the structure-reactivity relationships of TET/AlkBH-enzymes in epigenetic modifications ultimately resulting in a better understanding of the level of control by the catalytic domain in these transformations.

Our first paper on this topic is out already:
Reactivity of a TET model complex against nucleobases: N. S. W. Jonasson, L. J. Daumann, Chem. Eur. J. 2019, 25, 12091. (Cover)



