Search
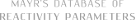
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
n-propylamine (in MeCN)  |
MeCN | N Param.: 15.11 sN Param.: 0.63 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
4-pyridone anion (in MeCN)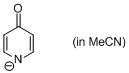  |
MeCN | N Param.: 20.22 sN Param.: 0.49 | J. Am. Chem. Soc. 2010, 132, 15380-15389 10.1021/ja106962u | |
(S,E)-2,2,3-trimethyl-4-oxo-5-((perfluorophenyl)methyl)-1-((E)-3-phenylallylidene)imidazolidin-1-ium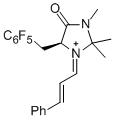  |
dichloromethane | E Param.: -6.00 | Angew. Chem. Int. Ed. 2013, 52, 7967-7971 10.1002/anie.201301864 | |
1-(phenylmethylamino)cyclohexene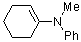  |
dichloromethane | N Param.: 10.73 sN Param.: 0.81 | Chem. Eur. J. 2003, 9, 2209-2218 10.1002/chem.200204666 | |
2-methyl-benzimidazole (in DMSO)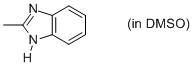  |
DMSO | N Param.: 10.02 sN Param.: 0.85 | Org. Biomol. Chem. 2010, 8, 1929-1935 10.1039/c000965b | |
(4-CF3)2-tritylium ion  |
E Param.: 2.28 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
1,1-dimethyl-2-methylenehydrazine (in CH2Cl2)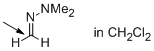  |
dichloromethane | N Param.: 6.98 sN Param.: 0.85 | Angew. Chem. Int. Ed. 2013, 52, 11900-11904 10.1002/anie.201305092 | |
2-(trimethylsilyl)propene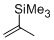  |
dichloromethane | N Param.: -1.46 sN Param.: 1.05 | Chem. Eur. J. 2014, 20, 1103-1110 10.1002/chem.201303215 | |
alpha-(trimethylsilyl)styrene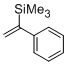  |
dichloromethane | N Param.: -1.13 sN Param.: 1.46 | Chem. Eur. J. 2014, 20, 1103-1110 10.1002/chem.201303215 | |
1-ethinylcyclohexenyl-Co2(CO)6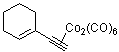  |
dichloromethane | N Param.: -0.44 sN Param.: 1.06 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y | |
anion of phenylnitromethane (in 91M9AN)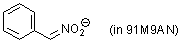  |
MeOH-MeCN mix | N Param.: 12.51 sN Param.: 0.67 | Eur. J. Org. Chem. 2006, , 2530-2537 10.1002/ejoc.200500769 | |
1% water/99% HFIP (w/w)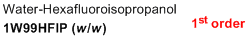  |
water-HFIP mix | N Param.: -1.93 sN Param.: 1.09 | J. Phys. Org. Chem. 2013, 26, 59-63 10.1002/poc.3064 | |
ethyl (E)-3-(dimethylamino)acrylate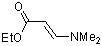  |
dichloromethane | N Param.: 9.43 sN Param.: 0.80 | Chem. Eur. J. 2003, 9, 2209-2218 10.1002/chem.200204666 | |
perhydroazepine (in water)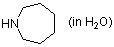  |
water | N Param.: 18.29 sN Param.: 0.46 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
thymine anion (in DMSO)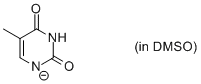  |
DMSO | N Param.: 17.63 sN Param.: 0.62 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
diethyl(methyl)silane  |
dichloromethane | N Param.: 3.40 sN Param.: 0.73 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
borane-4-methoxypyridine-complex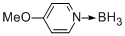  |
dichloromethane | N Param.: 11.01 sN Param.: 0.75 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
beta-(trimethylsilyl)styrene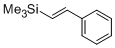  |
dichloromethane | N Param.: -0.43 sN Param.: 1.06 | Chem. Eur. J. 2014, 20, 1103-1110 10.1002/chem.201303215 | |
4-(dimethylamino)pyridine (in H2O)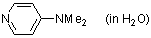  |
water | N Param.: 13.19 sN Param.: 0.56 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
p-toluidine (in MeCN)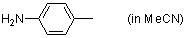  |
MeCN | N Param.: 13.19 sN Param.: 0.69 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z |
News
- 01/30/25:
Sesamol-derived ortho-quinone methides (o-QM) have been added (ChemEurJ 2024, e202403785 & OBC 2025, 23, 827-834) - 05/22/24:
Lactone enolates have been added (JOC 2024, 89, 6915-6928). - 11/13/24:
2-Methylene-1,2-dihydropyridines (2-pyNHOs) have been added (EurJOC 2024, 27, e202400373). - 05/21/24:
Mesoionic pyridinium-derived N-heterocyclic olefins (py-mNHOs) have been added (Angew. Chem. Int. Ed. 2024, 63, e202318283). - 10/19/23:
Enamines derived from 4-(alkylthio)-3-imidazolines and aldehydes have been added (ChemEurJ 2023, doi: 10.1002/chem.202302764). - 09/28/23:
Mesoionic N-heterocyclic olefins (mNHOs) have been added (Angew. Chem. Int. Ed. 2023, 62, e202309790). - 07/05/23:
S-Cyanomethylated imidazolidine-4-thione-derived enamines have been added (ChemComm 2023, 59, 8091). - 03/16/23:
Cyclic alpha-diazo carbonyl compounds added (EurJOC 2023, 26, e202300005) - 01/30/23:
NADH and NADPH reactivities (in water, pH 7) determined by Mayer and Moran have been added (OBC 2022, 21, 85-88). - 09/20/22:
Alkenyl boronate nucleophilicity by Liu, Ready and co-workers has been added (JACS 2022, 144, 16118). - 01/30/23:
Electrophilicities of diazo compounds in azo couplings (ChemEurJ 2022, 28, e202201376) - 01/30/23:
Diazocyclopentadiene added (Synthesis 2023, 55, 354-358) - 01/30/23:
Electrophilic indoles added (ChemCommun 2021, 57, 10071-10074) - 04/13/21:
Cyclic Michael acceptors added (Chem. Sci. 2021, 12, 4850-4865) - 05/21/21:
Thiophenolates (in DMSO) added (JOC 2021, 86, 5965-5972) - 05/06/20:
Heteroallenes added (JACS 2020, 142, 8383-8402). - 01/27/20:
Pyrrolidines and Imidazolidinones added (JACS 2020, 142, 1526-1547). - 05/06/20:
GSH reactivity added (Angew. Chem. Int. Ed. 2019, 58, 17704-17708) - 01/30/23:
Phenolates added (JOC 2019, 84, 8837-8858) - 04/24/18:
Ketones added (JACS 2018, 140, 5500). - 11/03/17:
Allyl-boron nucleophiles added (JACS 2017, 139, 15324) - 04/23/18:
Peroxide anions added (Angew. Chem. Int. Ed. 2017, 56, 13279). - 04/23/18:
Michael acceptors added (JACS 2017, 139, 13318). - 06/16/17:
Nucleophilicities of VNS reagents (JOC 2017, 82, 1011).
