C-Nucleophiles
20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28
« Back Forward » Found 602 molecules, displaying page 24 of 31 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
potassium trifluoro(1-methyl-1H-indol-2-yl)borate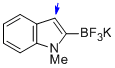  |
MeCN | N Param.: 9.55 sN Param.: 1.16 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(1-methyl-1H-indol-5-yl)borate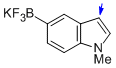  |
MeCN | N Param.: 8.77 sN Param.: 1.09 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-2-yl)trifluoroborate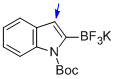  |
MeCN | N Param.: 6.46 sN Param.: 0.96 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-5-methoxy-1H-indol-2-yl)trifluoroborate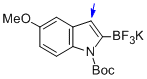  |
MeCN | N Param.: 7.10 sN Param.: 1.18 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-3-yl)trifluoroborate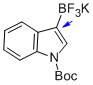  |
MeCN | N Param.: 4.06 sN Param.: 0.79 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
tert-butyl indole-1-carboxylate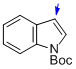  |
MeCN | N Param.: 1.68 sN Param.: 1.26 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(furan-2-yl)borate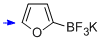  |
MeCN | N Param.: 5.99 sN Param.: 0.79 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(furan-3-yl)borate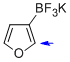  |
MeCN | N Param.: 6.83 sN Param.: 0.93 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium trifluoro(3-methoxy-thiophen-2-yl)borate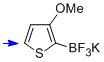  |
MeCN | N Param.: 7.32 sN Param.: 0.90 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
3-methoxythiophene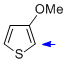  |
MeCN | N Param.: 3.06 sN Param.: 1.19 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
2-ethoxy-2-oxo-1-(pyridin-1-ium-1-yl)ethan-1-ide (in DMSO)  |
DMSO | N Param.: 26.71 sN Param.: 0.37 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
2-(diethylamino)-2-oxo-1-(pyridin-1-ium-1-yl)ethan-1-ide (in DMSO)  |
DMSO | N Param.: 27.45 sN Param.: 0.38 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
cyano(pyridin-1-ium-1-yl)methanide (in DMSO)  |
DMSO | N Param.: 25.94 sN Param.: 0.42 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
2-oxo-1-(pyridin-1-ium-1-yl)propan-1-ide (in DMSO)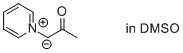  |
DMSO | N Param.: 20.24 sN Param.: 0.60 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
2-oxo-2-phenyl-1-(pyridin-1-ium-1-yl)ethan-1-ide (in DMSO)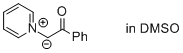  |
DMSO | N Param.: 19.46 sN Param.: 0.58 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
1-(4-(dimethylamino)pyridin-1-ium-1-yl)-2-oxo-2-phenylethan-1-ide (in DMSO)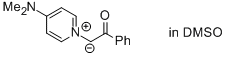  |
DMSO | N Param.: 21.61 sN Param.: 0.58 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
1-(3-chloropyridin-1-ium-1-yl)-2-oxo-2-phenylethan-1-ide (in DMSO)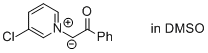  |
DMSO | N Param.: 17.98 sN Param.: 0.63 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
2-oxo-2-phenyl-1-(quinolin-1-ium-1-yl)ethan-1-ide (in DMSO)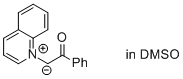  |
DMSO | N Param.: 19.38 sN Param.: 0.50 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
1-(isoquinolin-2-ium-2-yl)-2-oxo-2-phenylethan-1-ide (in DMSO)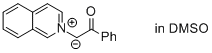  |
DMSO | N Param.: 20.08 sN Param.: 0.57 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
(E)-1-styrylpyrrolidine (in CH2Cl2)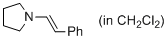  |
dichloromethane | N Param.: 12.26 sN Param.: 0.93 | J. Am. Chem. Soc. 2014, 136, 14263-14269 10.1021/ja508065e |
