N-Nucleophiles
7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15
« Back Forward » Found 353 molecules, displaying page 11 of 18 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
(2S,5S)-5-benzyl-3-methyl-2-(5-methylfuran-2-yl)imidazolidin-4-one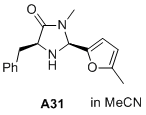  |
MeCN | N Param.: 8.76 sN Param.: 0.89 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(2R,5S)-5-benzyl-3-methyl-2-(5-methylfuran-2-yl)imidazolidin-4-one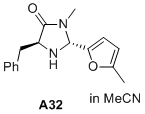  |
MeCN | N Param.: 7.39 sN Param.: 1.00 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
TCAP (in MeCN)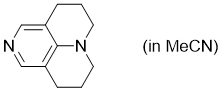  |
MeCN | N Param.: 15.60 sN Param.: 0.68 | Chem. Eur. J. 2013, 19, 6435-6442 10.1002/chem.201204452 | |
4-pyrrolidinopyridine (in MeCN)  |
MeCN | N Param.: 14.99 sN Param.: 0.69 | Chem. Eur. J. 2013, 19, 6435-6442 10.1002/chem.201204452 | |
pyridine (in MeCN)  |
MeCN | N Param.: 13.60 sN Param.: 0.60 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
2-methylpyridine (in MeCN)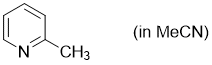  |
MeCN | N Param.: 10.98 sN Param.: 0.66 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
2,6-dimethylpyridine (in MeCN)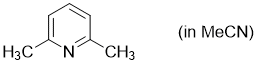  |
MeCN | N Param.: 9.11 sN Param.: 0.69 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
2,4,6-trimethylpyridine (in MeCN)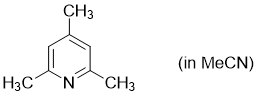  |
MeCN | N Param.: 9.39 sN Param.: 0.60 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in MeCN)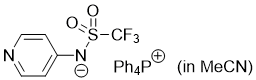  |
MeCN | N Param.: 16.38 sN Param.: 0.60 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
((4-methoxyphenyl)sulfonyl)(pyridin-4-yl)amide (Ph4P+) (in MeCN)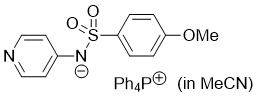  |
MeCN | N Param.: 17.28 sN Param.: 0.65 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
pyridin-4-yl((4-(trifluoromethyl)phenyl)sulfonyl)amide (Ph4P+) (in MeCN)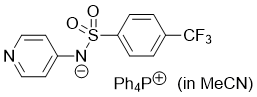  |
MeCN | N Param.: 19.51 sN Param.: 0.47 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl(tosyl)amide (Ph4P+) (in MeCN)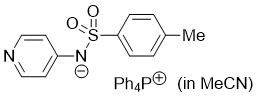  |
MeCN | N Param.: 17.19 sN Param.: 0.64 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4P+) (in MeCN)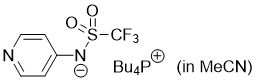  |
MeCN | N Param.: 16.48 sN Param.: 0.60 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4N+) (in MeCN)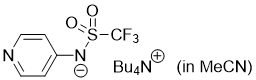  |
MeCN | N Param.: 16.68 sN Param.: 0.59 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
Se-HyperBTM (in MeCN)  |
MeCN | N Param.: 15.84 sN Param.: 0.57 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
azide ion (in 91iPr9AN)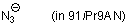  |
iPrOH-MeCN mix | N Param.: 17.07 sN Param.: 0.71 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
azide ion (in 91E9AN)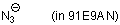  |
EtOH-MeCN mix | N Param.: 16.30 sN Param.: 0.73 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
2,2,2-trifluoroethylamine (in DMSO)  |
DMSO | N Param.: 12.15 sN Param.: 0.65 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
ethyl glycinate (in DMSO)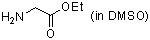  |
DMSO | N Param.: 14.30 sN Param.: 0.67 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
n-propylamine (in DMSO)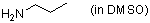  |
DMSO | N Param.: 15.70 sN Param.: 0.64 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y |
