N-Nucleophiles
10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18
« Back Forward » Found 343 molecules, displaying page 14 of 18 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
azide ion (in 91E9AN)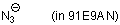  |
EtOH-MeCN mix | N Param.: 16.30 sN Param.: 0.73 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
1-methyl uracil anion (in DMSO)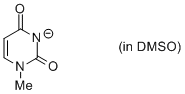  |
DMSO | N Param.: 16.36 sN Param.: 0.69 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in MeCN)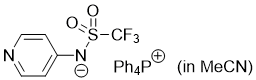  |
MeCN | N Param.: 16.38 sN Param.: 0.60 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
4-formyl-imidazole anion (in DMSO)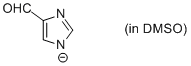  |
DMSO | N Param.: 16.40 sN Param.: 0.67 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
(R)-2-isopropylpyrrolidine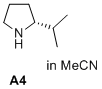  |
MeCN | N Param.: 16.44 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
hydrazine (in MeCN)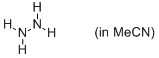  |
MeCN | N Param.: 16.45 sN Param.: 0.56 | Angew. Chem. Int. Ed. 2012, 51, 1353-1356 10.1002/anie.201107315 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4P+) (in MeCN)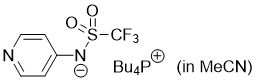  |
MeCN | N Param.: 16.48 sN Param.: 0.60 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
N-methyl-morpholine (in CH2Cl2)  |
dichloromethane | N Param.: 16.50 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
(R)-2-isopropyl-3,4-dihydro-2H-benzo[4,5]thiazolo[3,2-a]pyrimidine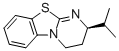  |
dichloromethane | N Param.: 16.50 sN Param.: 0.48 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
(S)-2-(methoxymethyl)pyrrolidine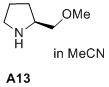  |
MeCN | N Param.: 16.50 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-benzhydrylpyrrolidine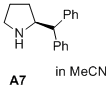  |
MeCN | N Param.: 16.61 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
Te-DHPB (in CH2Cl2)  |
dichloromethane | N Param.: 16.63 sN Param.: 0.65 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Te-HyperBTM (in DCM)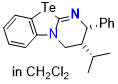  |
dichloromethane | N Param.: 16.63 sN Param.: 0.63 | ChemistryEurope 2026, 4, e202500443 10.1002/ceur.202500443 | |
2-Me super-dmap (in MeCN)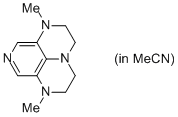  |
MeCN | N Param.: 16.65 sN Param.: 0.58 | Org. Lett. 2011, 13, 530-533 10.1021/ol1029589 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4N+) (in MeCN)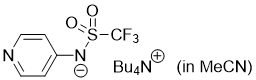  |
MeCN | N Param.: 16.68 sN Param.: 0.59 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
azide ion (in 91nPr9AN)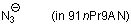  |
nPrOH-MeCN mix | N Param.: 16.70 sN Param.: 0.73 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
(S)-pyrrolidin-2-ylmethanol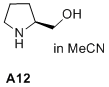  |
MeCN | N Param.: 16.74 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-methylpyrrolidine (in MeCN)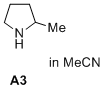  |
MeCN | N Param.: 16.78 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
N-methyl-morpholine (in MeCN)  |
MeCN | N Param.: 16.80 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
1-methyl-2-phenylpyrrolidine (in CH2Cl2)  |
dichloromethane | N Param.: 16.80 sN Param.: 0.49 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 |
