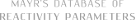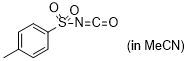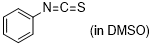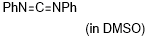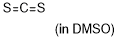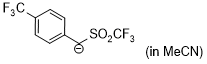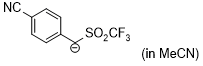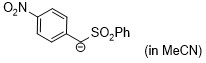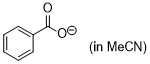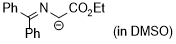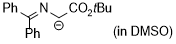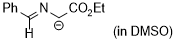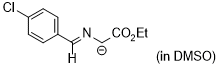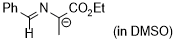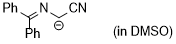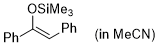| (E)-2,6-di-tert-butyl-4-(3-(4-methoxyphenyl)allylidene)cyclohexa-2,5-dien-1-one |
DMSO | E Param.: -17.42 |    | Org. Lett. 2020, 22, 2182-2186
10.1021/acs.orglett.[...] |
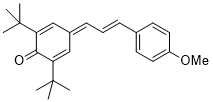
| 2,6-di-tert-butyl-4-(2,2,2-trifluoroethylidene)cyclohexa-2,5-dien-1-one |
DMSO | E Param.: -11.68 |    | Eur. J. Org. Chem. 2020, , 3812-3817
10.1002/ejoc.202000295 |
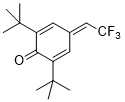
| phenyl isocyanate |
MeCN | E Param.: -15.38 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| p-tosyl isocyanate |
MeCN | E Param.: -7.69 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| phenyl isothiocyanate |
DMSO | E Param.: -18.15 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| p-nitrophenyl isothiocyanate |
DMSO | E Param.: -15.89 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| diphenylcarbodiimide |
DMSO | E Param.: -20.14 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| carbon disulfide |
DMSO | E Param.: -17.70 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| anion of 4-(trifluoromethyl)benzyl trifluoromethyl sulfone (in MeCN) |
MeCN | N Param.: 16.15 sN Param.: 0.99 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| anion of 4-cyanobenzyl trifluoromethyl sulfone (in MeCN) |
MeCN | N Param.: 15.62 sN Param.: 0.99 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
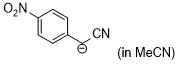
| anion of 4-nitrobenzyl-CN (in MeCN) |
MeCN | N Param.: 20.10 sN Param.: 0.71 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| anion of (4-NO2-C6H4)CH2SO2Ph (in MeCN) |
MeCN | N Param.: 19.90 sN Param.: 0.66 |    | J. Am. Chem. Soc. 2020, 142, 8383-8402
10.1021/jacs.0c01960 |
| benzoate (inMeCN) |
MeCN | N Param.: 16.45 sN Param.: 0.72 |    | J. Am. Chem. Soc. 2020, 142, 5221-5233
10.1021/jacs.9b12998 |
| 1-((diphenylmethylene)amino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 26.95 sN Param.: 0.52 |    | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 2-(tert-butoxy)-1-((diphenylmethylene)amino)-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 27.77 sN Param.: 0.47 |    | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 1-(benzylideneamino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 29.10 sN Param.: 0.50 |  | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 1-((4-chlorobenzylidene)amino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 29.02 sN Param.: 0.49 |   | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 2-(benzylideneamino)-1-ethoxy-1-oxopropan-2-ide (in DMSO) |
DMSO | N Param.: 30.82 sN Param.: 0.41 |   | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| cyano((diphenylmethylene)amino)methanide |
DMSO | N Param.: 29.50 sN Param.: 0.50 |  | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| (Z)-1,2-diphenyl-1-(trimethylsiloxy)ethene (in MeCN) |
MeCN | N Param.: 3.00 sN Param.: 0.83 |    | Synthesis 2019, 51, 1157-1170
10.1055/s-0037-1611634 |