Nucleophiles
46 | 47 | 48 | 49 | 50 | 51 | 52 | 53 | 54
« Back Forward » Found 1367 molecules, displaying page 50 of 69 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
hypochlorite (in water)  |
water | N Param.: 14.50 sN Param.: 0.58 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
imidazole (in 91M9AN)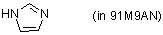  |
MeOH-MeCN mix | N Param.: 10.41 sN Param.: 0.70 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
imidazole (in DMSO)  |
DMSO | N Param.: 11.58 sN Param.: 0.79 | Org. Biomol. Chem. 2010, 8, 1929-1935 10.1039/c000965b | |
imidazole (in MeCN)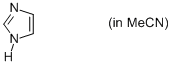  |
MeCN | N Param.: 11.47 sN Param.: 0.79 | Org. Biomol. Chem. 2010, 8, 1929-1935 10.1039/c000965b | |
imidazole (in water)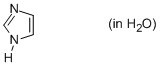  |
water | N Param.: 9.63 sN Param.: 0.57 | Org. Biomol. Chem. 2010, 8, 1929-1935 10.1039/c000965b | |
imidazole anion (in DMSO)  |
DMSO | N Param.: 21.09 sN Param.: 0.51 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
Indole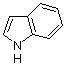  |
dichloromethane | N Param.: 5.55 sN Param.: 1.09 | J. Org. Chem. 2006, 71, 9088-9095 10.1021/jo0614339 | |
isobutylenyl-ethylether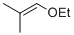  |
dichloromethane | N Param.: 4.23 sN Param.: 1.00 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
isopropanol (in aq. sulfuric acid)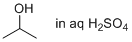  |
water | N Param.: -2.60 sN Param.: 0.80 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
isopropanol (in MeCN)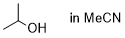  |
MeCN | N Param.: 6.82 sN Param.: 0.70 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
isopropanolate (in 91iPr9AN)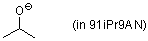  |
iPrOH-MeCN mix | N Param.: 17.71 sN Param.: 0.58 | Can. J. Chem. 2005, 83, 1554-1560 10.1139%2Fv05-170 | |
isopropanolate (in propan-2-ol)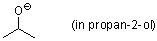  |
iPrOH | N Param.: 17.03 sN Param.: 0.63 | Can. J. Chem. 2005, 83, 1554-1560 10.1139%2Fv05-170 | |
isopropylamine (in MeCN)  |
MeCN | N Param.: 13.77 sN Param.: 0.70 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
isopropylamine (in water)  |
water | N Param.: 12.00 sN Param.: 0.56 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
KBH4 (in DMSO)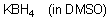  |
DMSO | N Param.: 15.14 sN Param.: 0.77 | Angew. Chem. Int. Ed. 2009, 48, 1958-1961 10.1002/anie.200804263 | |
leucine (anionic, in water)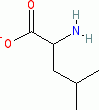  |
water | N Param.: 14.01 sN Param.: 0.52 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
lithium (5-methylfuran-2-yl)(4-(trifluoromethyl)phenyl)pinacolborate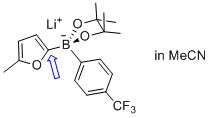  |
MeCN | N Param.: 8.13 sN Param.: 0.85 | Angew. Chem. Int. Ed. 2015, 54, 2780-2783 10.1002/anie.201410562 | |
lithium (5-methylthiophen-2-yl)(3,5-bis(trifluoromethyl)phenyl)glycolborate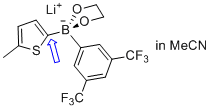  |
MeCN | N Param.: 11.23 sN Param.: 0.77 | Angew. Chem. Int. Ed. 2015, 54, 2780-2783 10.1002/anie.201410562 | |
lithium (5-methylthiophen-2-yl)(3,5-bis(trifluoromethyl)phenyl)neopentylglycolborate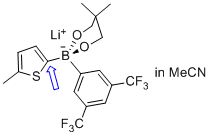  |
MeCN | N Param.: 10.13 sN Param.: 0.91 | Angew. Chem. Int. Ed. 2015, 54, 2780-2783 10.1002/anie.201410562 | |
lithium (5-methylthiophen-2-yl)(3,5-bis(trifluoromethyl)phenyl)pinacolborate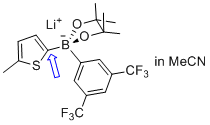  |
MeCN | N Param.: 5.53 sN Param.: 1.00 | Angew. Chem. Int. Ed. 2015, 54, 2780-2783 10.1002/anie.201410562 |
