Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
methylenecycloheptane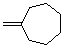  |
dichloromethane | N Param.: 2.24 sN Param.: 0.90 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
methylenecyclododecane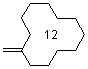  |
dichloromethane | N Param.: 1.52 sN Param.: 0.90 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
methylenecyclodecane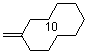  |
dichloromethane | N Param.: 2.82 sN Param.: 0.90 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
methylenecyclobutane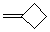  |
dichloromethane | N Param.: 1.65 sN Param.: 0.90 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
methyldiphenylsilane  |
dichloromethane | N Param.: 2.72 sN Param.: 0.73 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
methylamine (in water)  |
water | N Param.: 13.85 sN Param.: 0.53 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
methylamine (in MeCN)  |
MeCN | N Param.: 15.19 sN Param.: 0.68 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
methyl(phenyl)silane  |
dichloromethane | N Param.: 2.13 sN Param.: 0.73 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
methyl(neopentyl)(phenyl)silane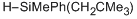  |
dichloromethane | N Param.: 0.87 sN Param.: 0.75 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
methyl L-prolinate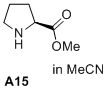  |
MeCN | N Param.: 14.75 sN Param.: 0.82 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
methyl glycinate (in water)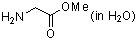  |
water | N Param.: 12.08 sN Param.: 0.60 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
methyl diazoacetate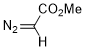  |
dichloromethane | N Param.: 4.68 sN Param.: 0.94 | J. Am. Chem. Soc. 2023, 145, 7416-7434 10.1021/jacs.2c13872 | |
methyl carbonate (in MeCN)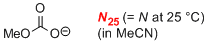  |
MeCN | N Param.: 16.03 sN Param.: 0.64 | Eur. J. Org. Chem. 2010, , 4205-4210 10.1002/ejoc.201000414 | |
methyl (cyclohexen-1-yl)prolinate (in MeCN)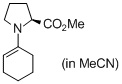  |
MeCN | N Param.: 14.96 sN Param.: 0.68 | Angew. Chem. Int. Ed. 2010, 49, 9526-9529 10.1002/anie.201004344 | |
methoxybis(phenylsulfonyl)methanide (in DMSO)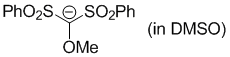  |
DMSO | N Param.: 17.29 sN Param.: 0.70 | Angew. Chem. Int. Ed. 2016, 55, 12845-12848 10.1002/anie.201605616 | |
methionine (anionic, in water)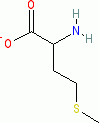  |
water | N Param.: 13.16 sN Param.: 0.58 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
methanolate (in methanol)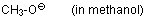  |
MeOH | N Param.: 15.78 sN Param.: 0.56 | Can. J. Chem. 2005, 83, 1554-1560 10.1139%2Fv05-170 | |
methanolate (in 91M9AN)  |
MeOH-MeCN mix | N Param.: 14.51 sN Param.: 0.68 | Can. J. Chem. 2005, 83, 1554-1560 10.1139%2Fv05-170 | |
methanol (in MeCN)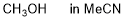  |
MeCN | N Param.: 6.86 sN Param.: 0.73 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
methanol  |
MeOH | N Param.: 7.54 sN Param.: 0.92 | J. Am. Chem. Soc. 2004, 126, 5174-5181 10.1021/ja031828z | |
methanesulfonamide anion (in DMSO)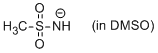  |
DMSO | N Param.: 18.61 sN Param.: 0.53 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
MeSO2-CH-CO2Et (in DMSO)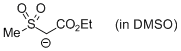  |
DMSO | N Param.: 18.00 sN Param.: 0.66 | Chem. Eur. J. 2010, 16, 8610-8614 10.1002/chem.201001455 | |
MeSO-CH-CO2Et (in DMSO)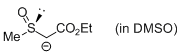  |
DMSO | N Param.: 20.61 sN Param.: 0.64 | Chem. Eur. J. 2010, 16, 8610-8614 10.1002/chem.201001455 | |
MeO-Breslow 1e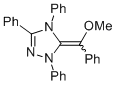  |
THF | N Param.: 15.65 sN Param.: 0.52 | Angew. Chem. Int. Ed. 2012, 51, 10408-10412 10.1002/anie.201204524 | |
MeO-Breslow 1c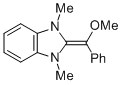  |
THF | N Param.: 16.61 sN Param.: 0.68 | Angew. Chem. Int. Ed. 2012, 51, 10408-10412 10.1002/anie.201204524 | |
MeO-Breslow 1b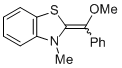  |
THF | N Param.: 10.45 sN Param.: 0.81 | Angew. Chem. Int. Ed. 2012, 51, 10408-10412 10.1002/anie.201204524 | |
MeO-Breslow 1a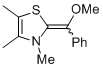  |
THF | N Param.: 14.77 sN Param.: 0.80 | Angew. Chem. Int. Ed. 2012, 51, 10408-10412 10.1002/anie.201204524 | |
Meldrum's acid iodonium ylide (in CH2Cl2)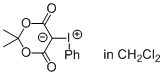  |
dichloromethane | N Param.: 4.36 sN Param.: 1.06 | J. Am. Chem. Soc. 2016, 138, 10304-10313 10.1021/jacs.6b05768 | |
Me2S=CH-CO2Et (in DMSO)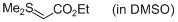  |
DMSO | N Param.: 15.85 sN Param.: 0.61 | Chem. Eur. J. 2010, 16, 8610-8614 10.1002/chem.201001455 | |
Me2S=CH-CN (in DMSO)  |
DMSO | N Param.: 16.23 sN Param.: 0.60 | Chem. Eur. J. 2010, 16, 8610-8614 10.1002/chem.201001455 | |
Me2S=CH(p-NO2-C6H4) (in DMSO)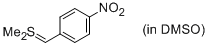  |
DMSO | N Param.: 18.42 sN Param.: 0.65 | J. Am. Chem. Soc. 2010, 132, 17894-17900 10.1021/ja1084749 | |
Me2S=CH(4-CN-C6H4) (in DMSO)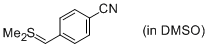  |
DMSO | N Param.: 21.07 sN Param.: 0.68 | J. Am. Chem. Soc. 2010, 132, 17894-17900 10.1021/ja1084749 | |
Me2S(O)=CH2 (in DMSO)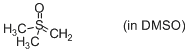  |
DMSO | N Param.: 21.29 sN Param.: 0.47 | J. Am. Chem. Soc. 2010, 132, 17894-17900 10.1021/ja1084749 | |
Me2 Imd boronate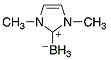  |
dichloromethane | N Param.: 11.88 sN Param.: 0.71 | Org. Lett. 2012, 14, 82-85 10.1021/ol202836p | |
maleimide anion (in DMSO)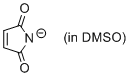  |
DMSO | N Param.: 14.87 sN Param.: 0.76 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
magnesium 2-carboperoxybenzoate (in H2O)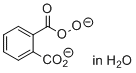  |
water | N Param.: 18.43 sN Param.: 0.46 | Angew. Chem. Int. Ed. 2017, 56, 13279-13282 10.1002/anie.201707086 | |
m-xylene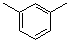  |
dichloromethane | N Param.: -3.57 sN Param.: 2.08 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y | |
m-nitrophenolate (in MeCN)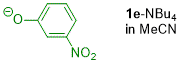  |
MeCN | N Param.: 25.51 sN Param.: 0.43 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-nitrophenolate (in DMSO)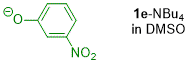  |
DMSO | N Param.: 21.29 sN Param.: 0.54 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-nitrophenolate (in DMF)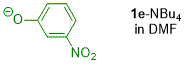  |
DMF | N Param.: 22.41 sN Param.: 0.54 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-methoxyphenolate (in MeCN)  |
MeCN | N Param.: 18.81 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in MeCN)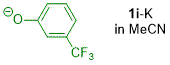  |
MeCN | N Param.: 23.20 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in DMSO)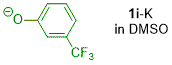  |
DMSO | N Param.: 22.62 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in DMF)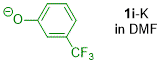  |
DMF | N Param.: 23.40 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
lithium indenide (in DMSO)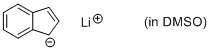  |
DMSO | N Param.: 23.66 sN Param.: 0.70 | Angew. Chem. Int. Ed. 2015, 54, 12497-12500 10.1002/anie.201501385 | |
lithium bis(5-methylthiophen-2-yl)pinacolborate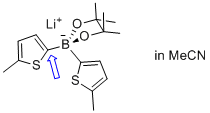  |
MeCN | N Param.: 7.67 sN Param.: 0.87 | Angew. Chem. Int. Ed. 2015, 54, 2780-2783 10.1002/anie.201410562 | |
lithium 5,5-dimethyl-2-(1-phenylethyl)-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborinan-2-uide (in MeCN)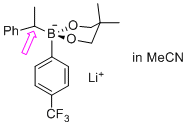  |
MeCN | N Param.: 11.29 sN Param.: 0.76 | Org. Lett. 2015, 17, 2614-2617 10.1021/acs.orglett.[...] | |
lithium 4,4,5,5-tetramethyl-2-phenyl-2-(prop-1-en-2-yl)-1,3,2-dioxaborolan-2-uide (in MeCN)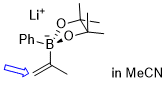  |
MeCN | N Param.: 11.00 sN Param.: 0.57 | J. Am. Chem. Soc. 2022, 144, 16118-16130 10.1021/jacs.2c06493 | |
lithium 4,4,5,5-tetramethyl-2-phenyl-2-(1-phenylethyl)-1,3,2-dioxaborolan-2-uide (in MeCN)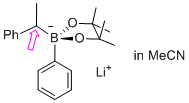  |
MeCN | N Param.: 10.97 sN Param.: 0.63 | Org. Lett. 2015, 17, 2614-2617 10.1021/acs.orglett.[...] | |
lithium 4,4,5,5-tetramethyl-2-(1-phenylethyl)-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborolan-2-uide (in MeCN)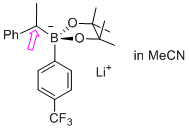  |
MeCN | N Param.: 8.56 sN Param.: 0.76 | Org. Lett. 2015, 17, 2614-2617 10.1021/acs.orglett.[...] |
