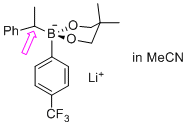
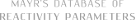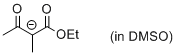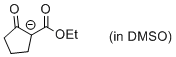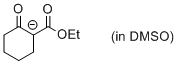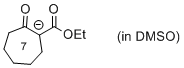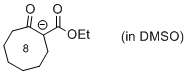| lithium 2-isopropyl-4,4,5,5-tetramethyl-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborolan-2-uide (in MeCN) |
MeCN | N Param.: 6.63 sN Param.: 0.75 |  | Org. Lett. 2015, 17, 2614-2617
10.1021/acs.orglett.[...] |
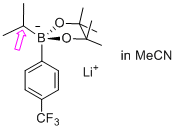
| lithium 2-cyclopropyl-4,4,5,5-tetramethyl-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborolan-2-uide (in MeCN) |
MeCN | N Param.: 7.72 sN Param.: 0.75 |  | Org. Lett. 2015, 17, 2614-2617
10.1021/acs.orglett.[...] |
| lithium 2-ethyl-4,4,5,5-tetramethyl-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborolan-2-uide (in MeCN) |
MeCN | N Param.: 7.68 sN Param.: 0.78 |    | Org. Lett. 2015, 17, 2614-2617
10.1021/acs.orglett.[...] |
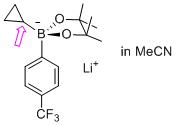
| lithium 5,5-dimethyl-2-(1-phenylethyl)-2-(4-(trifluoromethyl)phenyl)-1,3,2-dioxaborinan-2-uide (in MeCN) |
MeCN | N Param.: 11.29 sN Param.: 0.76 |    | Org. Lett. 2015, 17, 2614-2617
10.1021/acs.orglett.[...] |
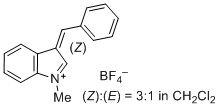
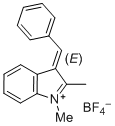
| 3-benzylidene-1-methyl-3H-indol-1-ium ion |
| E Param.: -1.80 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
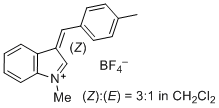
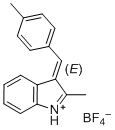
| 1-methyl-3-(4-methylbenzylidene)-3H-indol-1-ium ion |
| E Param.: -2.19 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
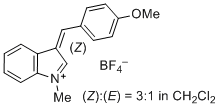
| 3-(4-methoxybenzylidene)-1-methyl-3H-indol-1-ium iom |
| E Param.: -3.02 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
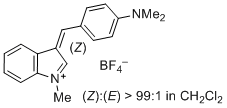
| 3-(4-(dimethylamino)benzylidene)-1-methyl-3H-indol-1-ium ion |
| E Param.: -6.26 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
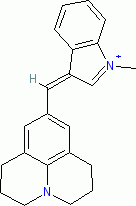
| 1-methyl-3-((julolidin-9-yl)methylene)-3H-indol-1-ium ion |
| E Param.: -7.79 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
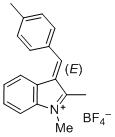
| (E)-2-methyl-3-(4-methylbenzylidene)-3H-indol-1-ium ion |
| E Param.: -5.15 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
| (E)-1,2-dimethyl-3-(4-methylbenzylidene)-3H-indol-1-ium ion |
| E Param.: -5.05 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
| (E)-3-benzylidene-1,2-dimethyl-3H-indol-1-ium ion |
| E Param.: -4.96 |   | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
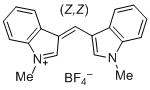
| bis(1-methylindol-3-yl)methylium ion |
| E Param.: -5.99 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
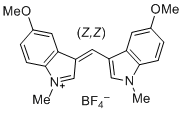
| bis(5-methoxy-1-methylindol-3-yl)methylium ion |
| E Param.: -6.90 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
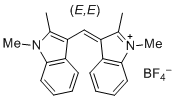
| bis(1,2-dimethylindol-3-yl)methylium ion |
| E Param.: -10.23 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
| 1-ethoxy-2-methyl-1,3-dioxobutan-2-ide (in DMSO) |
DMSO | N Param.: 19.58 sN Param.: 0.73 |    | Eur. J. Org. Chem. 2015, , 7594-7601
10.1002/ejoc.201501107 |
| 1-(ethoxycarbonyl)-2-oxocyclopentan-1-ide (in DMSO) |
DMSO | N Param.: 18.63 sN Param.: 0.82 |    | Eur. J. Org. Chem. 2015, , 7594-7601
10.1002/ejoc.201501107 |
| 1-(ethoxycarbonyl)-2-oxocyclohexan-1-ide (in DMSO) |
DMSO | N Param.: 18.32 sN Param.: 0.84 |    | Eur. J. Org. Chem. 2015, , 7594-7601
10.1002/ejoc.201501107 |
| 1-(ethoxycarbonyl)-2-oxocycloheptan-1-ide |
DMSO | N Param.: 19.53 sN Param.: 0.83 |    | Eur. J. Org. Chem. 2015, , 7594-7601
10.1002/ejoc.201501107 |
| 1-(ethoxycarbonyl)-2-oxocyclooctan-1-ide (in DMSO) |
DMSO | N Param.: 20.02 sN Param.: 0.76 |    | Eur. J. Org. Chem. 2015, , 7594-7601
10.1002/ejoc.201501107 |