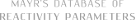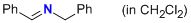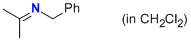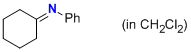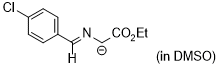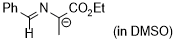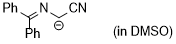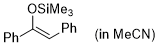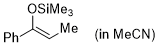| N-methyl-1-phenylmethanimine (in CH2Cl2) |
dichloromethane | N Param.: 8.60 sN Param.: 0.77 |    | Z. Naturforsch. B 2013, 68b, 693-699
10.5560/ZNB.2013-3085 |
| 1-phenyl-N-(phenylmethyl)methanimine (in CH2Cl2) |
dichloromethane | N Param.: 7.90 sN Param.: 0.76 |   | Z. Naturforsch. B 2013, 68b, 693-699
10.5560/ZNB.2013-3085 |
| N-phenylpropan-2-imine (in CH2Cl2) |
dichloromethane | N Param.: 9.53 sN Param.: 0.85 |    | Z. Naturforsch. B 2013, 68b, 693-699
10.5560/ZNB.2013-3085 |
| N-(phenylmethyl)propan-2-imine (in CH2Cl2) |
dichloromethane | N Param.: 11.13 sN Param.: 0.73 |    | Z. Naturforsch. B 2013, 68b, 693-699
10.5560/ZNB.2013-3085 |
| N-phenylcyclohexanimine (in CH2Cl2) |
dichloromethane | N Param.: 8.80 sN Param.: 1.00 |    | Z. Naturforsch. B 2013, 68b, 693-699
10.5560/ZNB.2013-3085 |
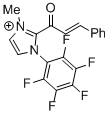
| 2-cinnamoyl-3-methyl-1-(perfluorophenyl)-1H-imidazol-3-ium |
DMSO | E Param.: -10.09 |    | Top. Catal. 2018, 61, 585-590
10.1007/s11244-018-0[...] |
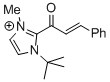
| 1-(tert-butyl)-2-cinnamoyl-3-methyl-1H-imidazol-3-ium |
DMSO | E Param.: -11.80 |    | Top. Catal. 2018, 61, 585-590
10.1007/s11244-018-0[...] |
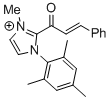
| 2-cinnamoyl-1-mesityl-3-methyl-1H-imidazol-3-ium |
DMSO | E Param.: -11.48 |    | Top. Catal. 2018, 61, 585-590
10.1007/s11244-018-0[...] |
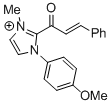
| 2-cinnamoyl-1-(4-methoxyphenyl)-3-methyl-1H-imidazol-3-ium |
DMSO | E Param.: -11.79 |    | Top. Catal. 2018, 61, 585-590
10.1007/s11244-018-0[...] |
| 2-cinnamoyl-1-(2,6-dimethoxyphenyl)-3-methyl-1H-imidazol-3-ium |
DMSO | E Param.: -12.02 |    | Top. Catal. 2018, 61, 585-590
10.1007/s11244-018-0[...] |
| 1-((diphenylmethylene)amino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 26.95 sN Param.: 0.52 |    | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 2-(tert-butoxy)-1-((diphenylmethylene)amino)-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 27.77 sN Param.: 0.47 |    | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 1-(benzylideneamino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 29.10 sN Param.: 0.50 |  | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 1-((4-chlorobenzylidene)amino)-2-ethoxy-2-oxoethan-1-ide (in DMSO) |
DMSO | N Param.: 29.02 sN Param.: 0.49 |   | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| 2-(benzylideneamino)-1-ethoxy-1-oxopropan-2-ide (in DMSO) |
DMSO | N Param.: 30.82 sN Param.: 0.41 |   | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| cyano((diphenylmethylene)amino)methanide |
DMSO | N Param.: 29.50 sN Param.: 0.50 |  | Tetrahedron 2019, 75, 459-463
10.1016/j.tet.2018.1[...] |
| (Z)-1,2-diphenyl-1-(trimethylsiloxy)ethene (in MeCN) |
MeCN | N Param.: 3.00 sN Param.: 0.83 |    | Synthesis 2019, 51, 1157-1170
10.1055/s-0037-1611634 |
| (Z)-1-phenyl-1-(trimethylsiloxy)propene (in MeCN) |
MeCN | N Param.: 5.18 sN Param.: 0.94 |    | Synthesis 2019, 51, 1157-1170
10.1055/s-0037-1611634 |
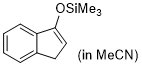
| 3-(trimethylsiloxy)-1H-indene (in MeCN) |
MeCN | N Param.: 7.32 sN Param.: 0.82 |    | Synthesis 2019, 51, 1157-1170
10.1055/s-0037-1611634 |
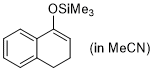
| 1-(trimethylsiloxy)-3,4-dihydronaphthalene (in MeCN) |
MeCN | N Param.: 5.06 sN Param.: 0.91 |    | Synthesis 2019, 51, 1157-1170
10.1055/s-0037-1611634 |