Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
potassium (1-(tert-butoxycarbonyl)-5-methoxy-1H-indol-2-yl)trifluoroborate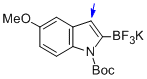  |
MeCN | N Param.: 7.10 sN Param.: 1.18 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-3-yl)trifluoroborate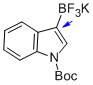  |
MeCN | N Param.: 4.06 sN Param.: 0.79 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
potassium (1-(tert-butoxycarbonyl)-1H-indol-2-yl)trifluoroborate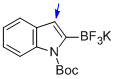  |
MeCN | N Param.: 6.46 sN Param.: 0.96 | J. Am. Chem. Soc. 2013, 135, 6317-6324 10.1021/ja4017655 | |
piperidine (in water)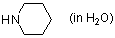  |
water | N Param.: 18.13 sN Param.: 0.44 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
piperidine (in MeCN)  |
MeCN | N Param.: 17.35 sN Param.: 0.68 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
piperidine (in DMSO)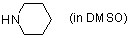  |
DMSO | N Param.: 17.19 sN Param.: 0.71 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
piperidine (in 91M9AN)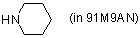  |
MeOH-MeCN mix | N Param.: 15.63 sN Param.: 0.64 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
piperidin-1-yl dithiocarbamate (in MeCN)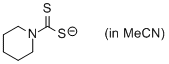  |
MeCN | N Param.: 23.84 sN Param.: 0.57 | Org. Biomol. Chem. 2011, 9, 8046-8050 10.1039/c1ob06245j | |
piperazine (in water)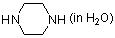  |
water | N Param.: 17.22 sN Param.: 0.50 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
phthalimidoperoxyhexanoate (in H2O)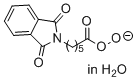  |
water | N Param.: 16.02 sN Param.: 0.54 | Angew. Chem. Int. Ed. 2017, 56, 13279-13282 10.1002/anie.201707086 | |
phthalimide anion (in DMSO)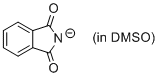  |
DMSO | N Param.: 15.52 sN Param.: 0.67 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
phosphaethynolate ion (in MeCN)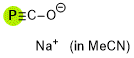  |
MeCN | N Param.: 19.02 sN Param.: 0.82 | Chem. Sci. 2024, 15, 14406-14414 10.1039/D4SC03518F | |
phenylsulfinate (in MeCN)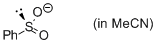  |
MeCN | N Param.: 20.11 sN Param.: 0.59 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phenylsulfinate (in DMSO)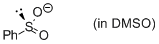  |
DMSO | N Param.: 19.60 sN Param.: 0.60 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phenylsulfinate (in 50W50AN)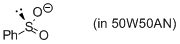  |
water-MeCN mix | N Param.: 13.75 sN Param.: 0.68 | J. Am. Chem. Soc. 2010, 132, 4796-4805 10.1021/ja9102056 | |
phenylsilane  |
dichloromethane | N Param.: 0.06 sN Param.: 0.71 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
phenyldiazomethane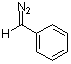  |
dichloromethane | N Param.: 9.35 sN Param.: 0.83 | Chem. Eur. J. 2003, 9, 4068-4076 10.1002/chem.200304913 | |
phenylalanine (anionic, in water)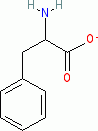  |
water | N Param.: 14.12 sN Param.: 0.53 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
phenylacetylene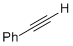  |
dichloromethane | N Param.: -0.04 sN Param.: 0.77 | Angew. Chem. Int. Ed. 2014, 53, 4968-4971 10.1002/anie.201402055 | |
phenolate (in MeCN)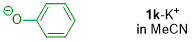  |
MeCN | N Param.: 18.53 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in DMSO)  |
DMSO | N Param.: 19.86 sN Param.: 0.71 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in DMF)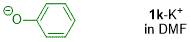  |
DMF | N Param.: 18.86 sN Param.: 0.89 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
Ph3P=CH-COPh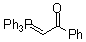  |
dichloromethane | N Param.: 9.54 sN Param.: 0.97 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph3P=CH-CO2Et (in DMSO)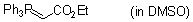  |
DMSO | N Param.: 12.21 sN Param.: 0.62 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph3P=CH-CO2Et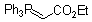  |
dichloromethane | N Param.: 12.79 sN Param.: 0.77 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph3P=CH-CN  |
dichloromethane | N Param.: 12.29 sN Param.: 0.75 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph3P=CH-CHO (in MeCN)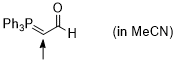  |
MeCN | N Param.: 9.09 sN Param.: 0.74 | J. Am. Chem. Soc. 2016, 138, 11272-11281 10.1021/jacs.6b06264 | |
Ph3P=CH-C(O)Me (in MeCN)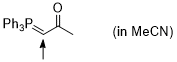  |
MeCN | N Param.: 10.27 sN Param.: 0.83 | J. Am. Chem. Soc. 2016, 138, 11272-11281 10.1021/jacs.6b06264 | |
Ph3P=C(CH3)-CO2Et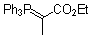  |
dichloromethane | N Param.: 13.09 sN Param.: 0.73 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph2P(O)CH(-)CO2Et (in DMSO)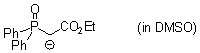  |
DMSO | N Param.: 19.20 sN Param.: 0.69 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
Ph2P(O)CH(-)CN (in DMSO)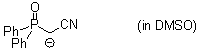  |
DMSO | N Param.: 18.69 sN Param.: 0.72 | J. Am. Chem. Soc. 2009, 131, 704-714 10.1021/ja8056216 | |
peroxybenzoate (in H2O)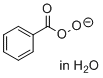  |
water | N Param.: 18.57 sN Param.: 0.43 | Angew. Chem. Int. Ed. 2017, 56, 13279-13282 10.1002/anie.201707086 | |
peroxy-GABA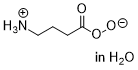  |
water | N Param.: 14.33 sN Param.: 0.57 | Eur. J. Org. Chem. 2018, , 6010-6017 10.1002/ejoc.201801158 | |
peroxy-beta-alanate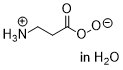  |
water | N Param.: 14.07 sN Param.: 0.56 | Eur. J. Org. Chem. 2018, , 6010-6017 10.1002/ejoc.201801158 | |
peroxomonosulfate (in water)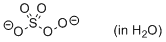  |
water | N Param.: 14.41 sN Param.: 0.60 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
perhydroazepine (in water)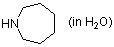  |
water | N Param.: 18.29 sN Param.: 0.46 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
p-tosylmethyl isocyanide (TosMIC)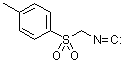  |
dichloromethane | N Param.: 3.50 sN Param.: 0.76 | Angew. Chem. Int. Ed. 2007, 46, 3563-3566 10.1002/anie.200605205 | |
p-toluidine (in water)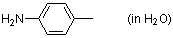  |
water | N Param.: 13.00 sN Param.: 0.79 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
p-toluidine (in MeCN)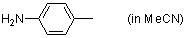  |
MeCN | N Param.: 13.19 sN Param.: 0.69 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
p-toluenesulfonamide anion (in DMSO)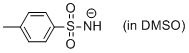  |
DMSO | N Param.: 17.14 sN Param.: 0.60 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
p-nitrophenolate (in DMSO)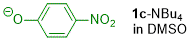  |
DMSO | N Param.: 14.32 sN Param.: 0.77 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-nitrobenzoate (in MeCN)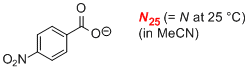  |
MeCN | N Param.: 15.30 sN Param.: 0.76 | J. Am. Chem. Soc. 2008, 130, 3012-3022 10.1021/ja0765464 | |
p-nitrobenzoate (in acetone)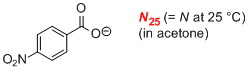  |
acetone | N Param.: 18.74 sN Param.: 0.68 | J. Am. Chem. Soc. 2008, 130, 3012-3022 10.1021/ja0765464 | |
p-methylphenolate (in H2O)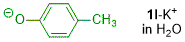  |
water | N Param.: 14.30 sN Param.: 0.55 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMSO)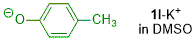  |
DMSO | N Param.: 19.08 sN Param.: 0.87 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMF)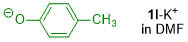  |
DMF | N Param.: 19.65 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in MeCN)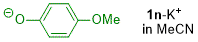  |
MeCN | N Param.: 20.62 sN Param.: 0.69 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMSO)  |
DMSO | N Param.: 20.09 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMF)  |
DMF | N Param.: 19.90 sN Param.: 0.82 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-cymene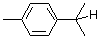  |
dichloromethane | N Param.: -2.80 sN Param.: 0.97 | J. Am. Chem. Soc. 2002, 124, 4076-4083 10.1021/ja0121538 |
