C-Nucleophiles
19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27
« Back Forward » Found 602 molecules, displaying page 23 of 31 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
barbiturate anion (in DMSO)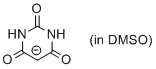  |
DMSO | N Param.: 15.59 sN Param.: 0.80 | J. Org. Chem. 2017, 82, 8476-8488 10.1021/acs.joc.7b01223 | |
benzyl (1-phenylvinyl)carbamate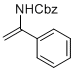  |
MeCN | N Param.: 6.21 sN Param.: 0.87 | Chem. Eur. J. 2012, 18, 5732-5740 10.1002/chem.201103519 | |
benzyl isocyanide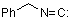  |
dichloromethane | N Param.: 4.90 sN Param.: 0.74 | Angew. Chem. Int. Ed. 2007, 46, 3563-3566 10.1002/anie.200605205 | |
beta-(trimethylsilyl)styrene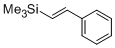  |
dichloromethane | N Param.: -0.43 sN Param.: 1.06 | Chem. Eur. J. 2014, 20, 1103-1110 10.1002/chem.201303215 | |
bis(4-nitrophenyl)methanide (in DMSO)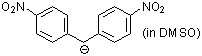  |
DMSO | N Param.: 19.92 sN Param.: 0.67 | ARKIVOC 2008, (x), 37-53 10.3998/ark.5550190.[...] | |
bis(phenylsulfonyl)methanide (in DMSO)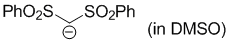  |
DMSO | N Param.: 15.68 sN Param.: 0.74 | Angew. Chem. Int. Ed. 2016, 55, 12845-12848 10.1002/anie.201605616 | |
bromo(phenylsulfonyl)methanide (in DMSO)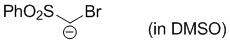  |
DMSO | N Param.: 23.90 sN Param.: 0.62 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
buta-1,3-diene  |
dichloromethane | N Param.: -0.87 sN Param.: 1.00 | Acc. Chem. Res. 2003, 36, 66-77 10.1021/ar020094c | |
chloro((4-chlorophenyl)sulfonyl)methanide (in DMSO)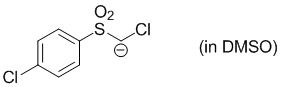  |
DMSO | N Param.: 26.90 sN Param.: 0.45 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro((4-cyanophenyl)sulfonyl)methanide (in DMSO)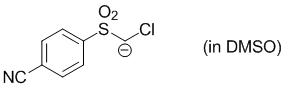  |
DMSO | N Param.: 25.59 sN Param.: 0.51 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro((4-nitrophenyl)sulfonyl)methanide (in DMSO)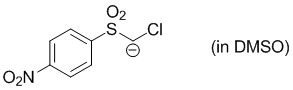  |
DMSO | N Param.: 24.88 sN Param.: 0.49 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro(phenylsulfonyl)methanide (in DMF)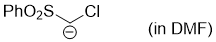  |
DMF | N Param.: 26.64 sN Param.: 0.64 | Chem. Eur. J. 2008, 14, 6108-6118 10.1002/chem.200800329 | |
chloro(phenylsulfonyl)methanide (in DMSO)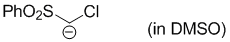  |
DMSO | N Param.: 28.27 sN Param.: 0.42 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
Cp2Ti(CH2Ph)2 - Dibenzyltitanocene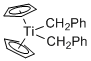  |
dichloromethane | N Param.: 4.38 sN Param.: 1.00 | Chem. Eur. J. 2016, 22, 11196-11200 10.1002/chem.201602452 | |
Cp2Zr(CH2Ph)2 - Dibenzylzirconocene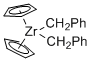  |
dichloromethane | N Param.: 5.10 sN Param.: 1.03 | Chem. Eur. J. 2016, 22, 11196-11200 10.1002/chem.201602452 | |
Cp2Zr(Me)2 - Dimethylzirconocene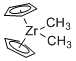  |
dichloromethane | N Param.: 4.35 sN Param.: 1.09 | Chem. Eur. J. 2016, 22, 11196-11200 10.1002/chem.201602452 | |
cyanide (in MeCN)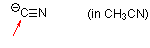  |
MeCN | N Param.: 16.27 sN Param.: 0.70 | Angew. Chem. Int. Ed. 2005, 44, 142-145 10.1002/anie.200461640 | |
cyano((diphenylmethylene)amino)methanide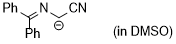  |
DMSO | N Param.: 29.50 sN Param.: 0.50 | Tetrahedron 2019, 75, 459-463 10.1016/j.tet.2018.1[...] | |
cyano(pyridin-1-ium-1-yl)methanide (in DMSO)  |
DMSO | N Param.: 25.94 sN Param.: 0.42 | J. Am. Chem. Soc. 2013, 135, 15216-15224 10.1021/ja407885h | |
cyclohepta-1,3,5-trienyl-Fe(CO)3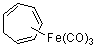  |
dichloromethane | N Param.: 3.42 sN Param.: 0.94 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y |
