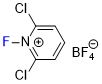
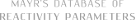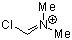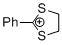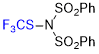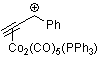| N-fluoro-2,6-dichloropyridinium tetrafluoroborate |
| E Param.: -5.29 |    | J. Am. Chem. Soc. 2018, 140, 11474-11486
10.1021/jacs.8b07147 |
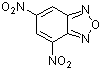
| 4,6-dinitrobenzofurazan |
| E Param.: -5.46 |    | J. Org. Chem. 2005, 70, 6242-6253
10.1021/jo0505526 |
| MacMillan-iminium ion (2nd generation) |
MeCN | E Param.: -5.52 |    | Angew. Chem. Int. Ed. 2011, 50, 9953-9956
10.1002/anie.201103683 |
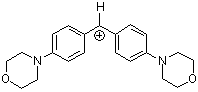
| (mor)2CH+ |
| E Param.: -5.53 |      | J. Am. Chem. Soc. 2001, 123, 9500-9512
10.1021/ja010890y |
| Vilsmeier ion |
| E Param.: -5.77 |  | Acc. Chem. Res. 2003, 36, 66-77
10.1021/ar020094c |
| 4-aza-6-nitrobenzofuroxan |
| E Param.: -5.86 |    | J. Org. Chem. 2005, 70, 6242-6253
10.1021/jo0505526 |
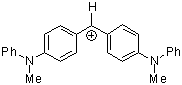
| (mpa)2CH+ |
| E Param.: -5.89 |      | J. Am. Chem. Soc. 2001, 123, 9500-9512
10.1021/ja010890y |
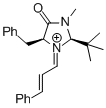
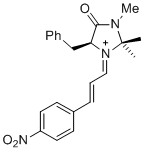
| (S,E)-5-benzyl-2,2,3-trimethyl-1-((E)-3-(4-nitrophenyl)allylidene)-4-oxoimidazolidin-1-ium |
dichloromethane | E Param.: -5.90 |   | Asian J. Org. Chem. 2014, 3, 550-555
10.1002/ajoc.201402009 |
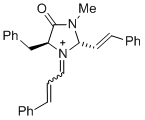
| (2S,5S)-5-benzyl-3-methyl-4-oxo-1-(3-phenylallylidene)-2-((E)-styryl)imidazolidin-1-ium |
dichloromethane | E Param.: -5.90 |   | Asian J. Org. Chem. 2014, 3, 550-555
10.1002/ajoc.201402009 |
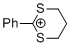
| 2-phenyl-1,3-dithiolan-2-ylium ion |
| E Param.: -5.91 |    | Acc. Chem. Res. 2003, 36, 66-77
10.1021/ar020094c |
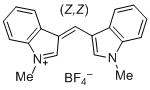
| bis(1-methylindol-3-yl)methylium ion |
| E Param.: -5.99 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
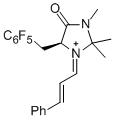
| (S,E)-2,2,3-trimethyl-4-oxo-5-((perfluorophenyl)methyl)-1-((E)-3-phenylallylidene)imidazolidin-1-ium |
dichloromethane | E Param.: -6.00 |   | Angew. Chem. Int. Ed. 2013, 52, 7967-7971
10.1002/anie.201301864 |
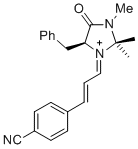
| (S,E)-5-benzyl-1-((E)-3-(4-cyanophenyl)allylidene)-2,2,3-trimethyl-4-oxoimidazolidin-1-ium |
dichloromethane | E Param.: -6.00 |   | Asian J. Org. Chem. 2014, 3, 550-555
10.1002/ajoc.201402009 |
| N-(phenylsulfonyl)-N-((trifluoromethyl)thio)benzenesulfonamide |
| E Param.: -6.06 |    | Angew. Chem. Int. Ed. 2018, 57, 12690-12695
10.1002/anie.201805859 |
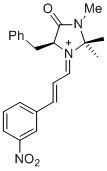
| (S,E)-5-benzyl-2,2,3-trimethyl-1-((E)-3-(3-nitrophenyl)allylidene)-4-oxoimidazolidin-1-ium |
dichloromethane | E Param.: -6.10 |   | Asian J. Org. Chem. 2014, 3, 550-555
10.1002/ajoc.201402009 |
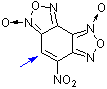
| 4-nitrobenzodifuroxan (NBDF) |
| E Param.: -6.15 |    | Chem. Eur. J. 2007, 13, 8317-8324
10.1002/chem.200700676 |
| 1-phenyl-propyn-1-ylium-Co2(CO)5(PPh3) |
| E Param.: -6.19 |    | Acc. Chem. Res. 2003, 36, 66-77
10.1021/ar020094c |
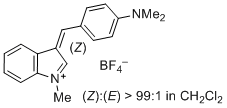
| 3-(4-(dimethylamino)benzylidene)-1-methyl-3H-indol-1-ium ion |
| E Param.: -6.26 |    | J. Org. Chem. 2015, 80, 8643-8656
10.1021/acs.joc.5b01298 |
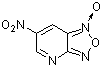
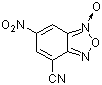
| 4-cyano-6-nitro-benzofuroxane |
| E Param.: -6.41 |    | J. Org. Chem. 2005, 70, 6242-6253
10.1021/jo0505526 |
| 2-phenyl-1,3-dithianylium ion |
| E Param.: -6.43 |    | Acc. Chem. Res. 2003, 36, 66-77
10.1021/ar020094c |