Nucleophiles
58 | 59 | 60 | 61 | 62 | 63 | 64 | 65 | 66
« Back Forward » Found 1367 molecules, displaying page 62 of 69 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
(S)-1-(3,5-bis(trifluoromethyl)phenyl)-3-(pyrrolidin-2-ylmethyl)thiourea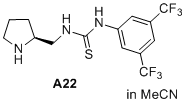  |
MeCN | N Param.: 14.97 sN Param.: 0.69 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(3,5-bis(trifluoromethyl)phenyl)-3-(pyrrolidin-2-ylmethyl)urea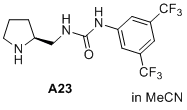  |
MeCN | N Param.: 17.50 sN Param.: 0.64 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-tritylpyrrolidine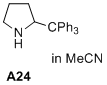  |
MeCN | N Param.: 9.16 sN Param.: 1.39 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(diphenyl((trimethylsilyl)oxy)methyl)pyrrolidine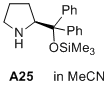  |
MeCN | N Param.: 12.03 sN Param.: 0.98 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-diphenyl(pyrrolidin-2-yl)methanol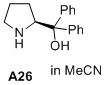  |
MeCN | N Param.: 16.18 sN Param.: 0.56 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(azidodiphenylmethyl)pyrrolidine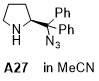  |
MeCN | N Param.: 9.90 sN Param.: 1.22 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-(triphenylsilyl)pyrrolidine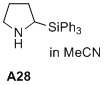  |
MeCN | N Param.: 14.00 sN Param.: 0.84 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-5-benzyl-2,2,3-trimethylimidazolidin-4-one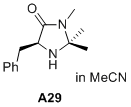  |
MeCN | N Param.: 6.04 sN Param.: 0.92 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(2S,5S)-5-benzyl-2-(tert-butyl)-3-methylimidazolidin-4-one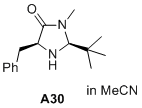  |
MeCN | N Param.: 5.44 sN Param.: 1.12 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(2S,5S)-5-benzyl-3-methyl-2-(5-methylfuran-2-yl)imidazolidin-4-one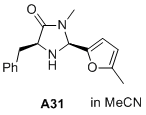  |
MeCN | N Param.: 8.76 sN Param.: 0.89 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(2R,5S)-5-benzyl-3-methyl-2-(5-methylfuran-2-yl)imidazolidin-4-one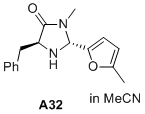  |
MeCN | N Param.: 7.39 sN Param.: 1.00 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
anion of 4-(trifluoromethyl)benzyl trifluoromethyl sulfone (in MeCN)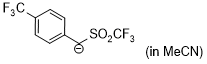  |
MeCN | N Param.: 16.15 sN Param.: 0.99 | J. Am. Chem. Soc. 2020, 142, 8383-8402 10.1021/jacs.0c01960 | |
anion of 4-cyanobenzyl trifluoromethyl sulfone (in MeCN)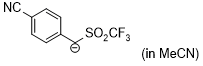  |
MeCN | N Param.: 15.62 sN Param.: 0.99 | J. Am. Chem. Soc. 2020, 142, 8383-8402 10.1021/jacs.0c01960 | |
anion of 4-nitrobenzyl-CN (in MeCN)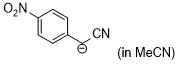  |
MeCN | N Param.: 20.10 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 8383-8402 10.1021/jacs.0c01960 | |
anion of (4-NO2-C6H4)CH2SO2Ph (in MeCN)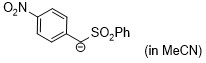  |
MeCN | N Param.: 19.90 sN Param.: 0.66 | J. Am. Chem. Soc. 2020, 142, 8383-8402 10.1021/jacs.0c01960 | |
benzoate (inMeCN)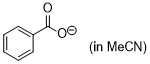  |
MeCN | N Param.: 16.45 sN Param.: 0.72 | J. Am. Chem. Soc. 2020, 142, 5221-5233 10.1021/jacs.9b12998 | |
dimethylsulfide (in CH2Cl2)  |
dichloromethane | N Param.: 12.32 sN Param.: 0.72 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
dimethylsulfide (in MeCN)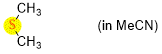  |
MeCN | N Param.: 12.70 sN Param.: 0.72 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
dimethylselenide (in CH2Cl2)  |
dichloromethane | N Param.: 12.60 sN Param.: 0.72 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 | |
dibutylsulfide (in CH2Cl2)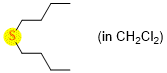  |
dichloromethane | N Param.: 11.86 sN Param.: 0.74 | Chem. Eur. J. 2021, 21, 11367-11376 10.1002/chem.202100977 |
