N-Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
n-propylamine (in MeCN)  |
MeCN | N Param.: 15.11 sN Param.: 0.63 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
methylamine (in MeCN)  |
MeCN | N Param.: 15.19 sN Param.: 0.68 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
4-aminopyridine (in CH2Cl2)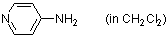  |
dichloromethane | N Param.: 15.20 sN Param.: 0.67 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
(S)-N-propylpyrrolidine-2-carboxamide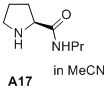  |
MeCN | N Param.: 15.20 sN Param.: 0.73 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-methyl-1,4,5,6-tetrahydropyrimidine (in CH2Cl2)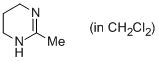  |
dichloromethane | N Param.: 15.21 sN Param.: 0.69 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
n-butylamine (in MeCN)  |
MeCN | N Param.: 15.27 sN Param.: 0.63 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
benzylamine (in DMSO)  |
DMSO | N Param.: 15.28 sN Param.: 0.65 | J. Am. Chem. Soc. 2009, 131, 11392-11401 10.1021/ja903207b | |
DBU (in MeCN)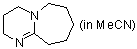  |
MeCN | N Param.: 15.29 sN Param.: 0.70 | Chem. Commun. 2008, , 1792-1794 10.1039/b801811a | |
(R)-2-benzhydryl-3,4-dihydro-2H-benzo[4,5]thiazolo[3,2-a]pyrimidine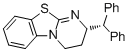  |
dichloromethane | N Param.: 15.30 sN Param.: 0.55 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
(S)-4-phenyl-1-(pyrrolidin-2-ylmethyl)-1H-1,2,3-triazole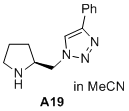  |
MeCN | N Param.: 15.32 sN Param.: 0.72 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-Ac super-dmap (in MeCN)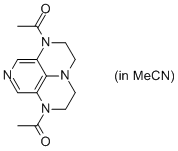  |
MeCN | N Param.: 15.39 sN Param.: 0.60 | Org. Lett. 2011, 13, 530-533 10.1021/ol1029589 | |
morpholine (in 91M9AN)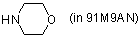  |
MeOH-MeCN mix | N Param.: 15.40 sN Param.: 0.64 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
(S)-2-(azidomethyl)pyrrolidine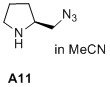  |
MeCN | N Param.: 15.43 sN Param.: 0.73 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
1-aminopropan-2-ol (in DMSO)  |
DMSO | N Param.: 15.47 sN Param.: 0.65 | J. Am. Chem. Soc. 2009, 131, 11392-11401 10.1021/ja903207b | |
DBN (in CH2Cl2)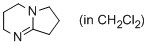  |
dichloromethane | N Param.: 15.50 sN Param.: 0.76 | ChemCatChem 2012, 4, 993-999 10.1002/cctc.201200143 | |
4-(dimethylamino)pyridine (in MeCN)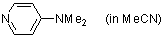  |
MeCN | N Param.: 15.51 sN Param.: 0.62 | J. Phys. Chem. A 2012, 116, 8494-8499 10.1021/jp3049247 | |
diethanolamine (in DMSO)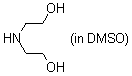  |
DMSO | N Param.: 15.51 sN Param.: 0.70 | J. Am. Chem. Soc. 2009, 131, 11392-11401 10.1021/ja903207b | |
phthalimide anion (in DMSO)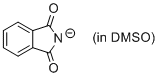  |
DMSO | N Param.: 15.52 sN Param.: 0.67 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
(S)-1-(pyrrolidin-2-ylmethyl)-1H-imidazole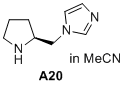  |
MeCN | N Param.: 15.55 sN Param.: 0.69 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
TCAP (in MeCN)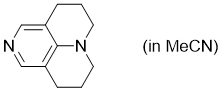  |
MeCN | N Param.: 15.60 sN Param.: 0.68 | Chem. Eur. J. 2013, 19, 6435-6442 10.1002/chem.201204452 | |
morpholine (in water)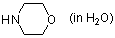  |
water | N Param.: 15.62 sN Param.: 0.54 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
piperidine (in 91M9AN)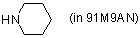  |
MeOH-MeCN mix | N Param.: 15.63 sN Param.: 0.64 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
morpholine (in MeCN)  |
MeCN | N Param.: 15.65 sN Param.: 0.74 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
2-(naphthylmethyl)quinuclidine (in MeCN)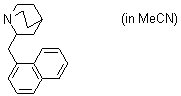  |
MeCN | N Param.: 15.66 sN Param.: 0.62 | J. Org. Chem. 2009, 74, 7157-7164 10.1021/jo901670w | |
N',N'-dimethylformohydrazide (in MeCN)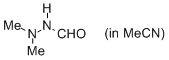  |
MeCN | N Param.: 15.69 sN Param.: 0.51 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
n-propylamine (in DMSO)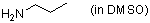  |
DMSO | N Param.: 15.70 sN Param.: 0.64 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
N-methyl-trifluoroacetamide anion (in DMSO)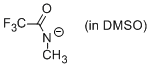  |
DMSO | N Param.: 15.70 sN Param.: 0.71 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
1-methyl-2-phenylpyrrolidine (in MeCN)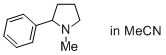  |
MeCN | N Param.: 15.70 sN Param.: 0.54 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
4-(dimethylamino)pyridine (in CH2Cl2)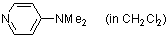  |
dichloromethane | N Param.: 15.80 sN Param.: 0.66 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
trifluoroacetamide anion (in DMSO)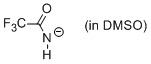  |
DMSO | N Param.: 15.81 sN Param.: 0.64 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
4-pyrrolidinopyridine (in CH2Cl2)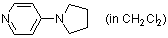  |
dichloromethane | N Param.: 15.90 sN Param.: 0.67 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
4-(dimethylamino)pyridine (in THF)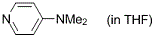  |
THF | N Param.: 15.90 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2011, 50, 6915-6919 10.1002/anie.201102435 | |
(S)-2-(pyrrolidin-2-ylmethyl)isoindoline-1,3-dione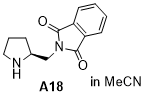  |
MeCN | N Param.: 15.90 sN Param.: 0.77 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
pyrrolidine (in 91M9AN)  |
MeOH-MeCN mix | N Param.: 15.97 sN Param.: 0.63 | Angew. Chem. Int. Ed. 2006, 45, 3869-3874 10.1002/anie.200600542 | |
ethyl N-acetylcarbamate anion (in DMSO)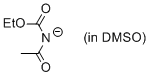  |
DMSO | N Param.: 15.99 sN Param.: 0.70 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
succinimide anion (in DMSO)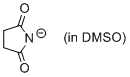  |
DMSO | N Param.: 16.03 sN Param.: 0.66 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
diacetamide anion (in DMSO)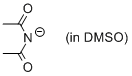  |
DMSO | N Param.: 16.05 sN Param.: 0.70 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
2-formyl-imidazole anion (in DMSO)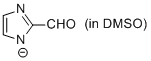  |
DMSO | N Param.: 16.06 sN Param.: 0.68 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
ethanolamine (in DMSO)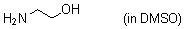  |
DMSO | N Param.: 16.07 sN Param.: 0.61 | J. Am. Chem. Soc. 2009, 131, 11392-11401 10.1021/ja903207b | |
DBU (in THF)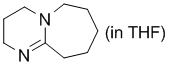  |
THF | N Param.: 16.12 sN Param.: 0.67 | Angew. Chem. Int. Ed. 2011, 50, 6915-6919 10.1002/anie.201102435 | |
1,2,3,5,6,7-hexahydroimidazo[1,2-a]pyrimidine (TBN)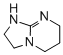  |
dichloromethane | N Param.: 16.15 sN Param.: 0.73 | ChemCatChem 2012, 4, 993-999 10.1002/cctc.201200143 | |
1,2-dimethylhydrazine (in MeCN)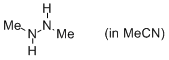  |
MeCN | N Param.: 16.15 sN Param.: 0.68 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
2,3,4,6,7,8-hexahydro-1H-pyrimido[1,2-a]pyrimidine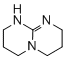  |
dichloromethane | N Param.: 16.16 sN Param.: 0.75 | ChemCatChem 2012, 4, 993-999 10.1002/cctc.201200143 | |
(S)-diphenyl(pyrrolidin-2-yl)methanol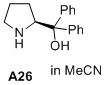  |
MeCN | N Param.: 16.18 sN Param.: 0.56 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
DBN (in MeCN)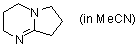  |
MeCN | N Param.: 16.28 sN Param.: 0.67 | Chem. Commun. 2008, , 1792-1794 10.1039/b801811a | |
benzotriazole anion (in DMSO)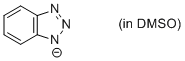  |
DMSO | N Param.: 16.29 sN Param.: 0.65 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
azide ion (in 91E9AN)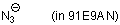  |
EtOH-MeCN mix | N Param.: 16.30 sN Param.: 0.73 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
1-methyl uracil anion (in DMSO)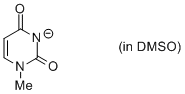  |
DMSO | N Param.: 16.36 sN Param.: 0.69 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in MeCN)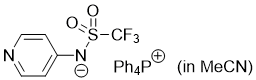  |
MeCN | N Param.: 16.38 sN Param.: 0.60 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
4-formyl-imidazole anion (in DMSO)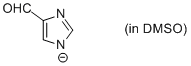  |
DMSO | N Param.: 16.40 sN Param.: 0.67 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
(R)-2-isopropylpyrrolidine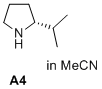  |
MeCN | N Param.: 16.44 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
hydrazine (in MeCN)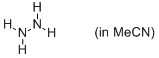  |
MeCN | N Param.: 16.45 sN Param.: 0.56 | Angew. Chem. Int. Ed. 2012, 51, 1353-1356 10.1002/anie.201107315 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4P+) (in MeCN)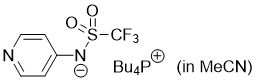  |
MeCN | N Param.: 16.48 sN Param.: 0.60 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
N-methyl-morpholine (in CH2Cl2)  |
dichloromethane | N Param.: 16.50 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
(R)-2-isopropyl-3,4-dihydro-2H-benzo[4,5]thiazolo[3,2-a]pyrimidine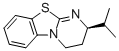  |
dichloromethane | N Param.: 16.50 sN Param.: 0.48 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
(S)-2-(methoxymethyl)pyrrolidine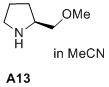  |
MeCN | N Param.: 16.50 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-benzhydrylpyrrolidine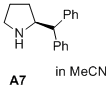  |
MeCN | N Param.: 16.61 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-Me super-dmap (in MeCN)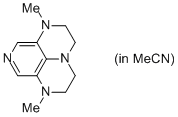  |
MeCN | N Param.: 16.65 sN Param.: 0.58 | Org. Lett. 2011, 13, 530-533 10.1021/ol1029589 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Bu4N+) (in MeCN)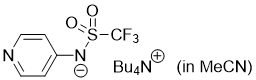  |
MeCN | N Param.: 16.68 sN Param.: 0.59 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
azide ion (in 91nPr9AN)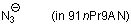  |
nPrOH-MeCN mix | N Param.: 16.70 sN Param.: 0.73 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
(S)-pyrrolidin-2-ylmethanol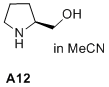  |
MeCN | N Param.: 16.74 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-methylpyrrolidine (in MeCN)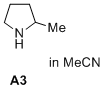  |
MeCN | N Param.: 16.78 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
N-methyl-morpholine (in MeCN)  |
MeCN | N Param.: 16.80 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
1-methyl-2-phenylpyrrolidine (in CH2Cl2)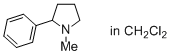  |
dichloromethane | N Param.: 16.80 sN Param.: 0.49 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
2-Et super-dmap (in MeCN)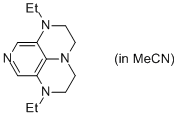  |
MeCN | N Param.: 16.81 sN Param.: 0.60 | Org. Lett. 2011, 13, 530-533 10.1021/ol1029589 | |
morpholine (in DMSO)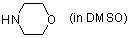  |
DMSO | N Param.: 16.96 sN Param.: 0.67 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
uracil anion (in DMSO)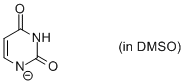  |
DMSO | N Param.: 17.04 sN Param.: 0.63 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
azide ion (in 91iPr9AN)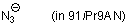  |
iPrOH-MeCN mix | N Param.: 17.07 sN Param.: 0.71 | J. Phys. Org. Chem. 2006, 19, 706-713 10.1002/poc.1063 | |
triethylamine (in MeCN)  |
MeCN | N Param.: 17.10 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
dimethylamine (in water)  |
water | N Param.: 17.12 sN Param.: 0.50 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
p-toluenesulfonamide anion (in DMSO)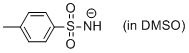  |
DMSO | N Param.: 17.14 sN Param.: 0.60 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
piperidine (in DMSO)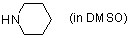  |
DMSO | N Param.: 17.19 sN Param.: 0.71 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | |
pyridin-4-yl(tosyl)amide (Ph4P+) (in MeCN)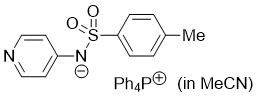  |
MeCN | N Param.: 17.19 sN Param.: 0.64 | J. Org. Chem. 2025, 90, 2298-2306 10.1021/acs.joc.4c02668 | |
nitrite ion (in MeCN)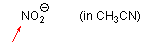  |
MeCN | N Param.: 17.20 sN Param.: 0.72 | Angew. Chem. Int. Ed. 2005, 44, 4623-4626 10.1002/anie.200501274 | |
pyrrolidine (in water)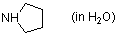  |
water | N Param.: 17.21 sN Param.: 0.49 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
piperazine (in water)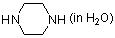  |
water | N Param.: 17.22 sN Param.: 0.50 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
methylhydrazine (in water)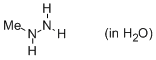  |
water | N Param.: 17.23 sN Param.: 0.45 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
(S)-pyrrolidin-2-ylmethanamine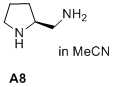  |
MeCN | N Param.: 17.24 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
((4-methoxyphenyl)sulfonyl)(pyridin-4-yl)amide (Ph4P+) (in MeCN)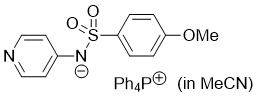  |
MeCN | N Param.: 17.28 sN Param.: 0.65 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
triethylamine (in CH2Cl2)  |
dichloromethane | N Param.: 17.30 sN Param.: 0.52 | J. Phys. Org. Chem. 2010, 23, 1029-1035 10.1002/poc.1707 | |
piperidine (in MeCN)  |
MeCN | N Param.: 17.35 sN Param.: 0.68 | Eur. J. Org. Chem. 2009, , 6379-6385 10.1002/ejoc.200900925 | |
(S)-N,N-dimethyl-1-(pyrrolidin-2-yl)methanamine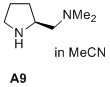  |
MeCN | N Param.: 17.41 sN Param.: 0.68 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-benzylpyrrolidine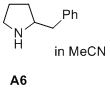  |
MeCN | N Param.: 17.43 sN Param.: 0.66 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(3,5-bis(trifluoromethyl)phenyl)-3-(pyrrolidin-2-ylmethyl)urea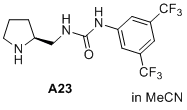  |
MeCN | N Param.: 17.50 sN Param.: 0.64 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
4,4-dimethyl-glutarimide anion (in DMSO)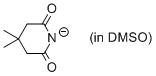  |
DMSO | N Param.: 17.52 sN Param.: 0.63 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
hydantoin anion (in DMSO)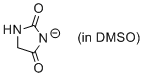  |
DMSO | N Param.: 17.52 sN Param.: 0.55 | J. Org. Chem. 2010, 75, 5250-5258 10.1021/jo1009883 | |
(S)-N,N-dimethylpyrrolidine-2-carboxamide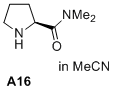  |
MeCN | N Param.: 17.61 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
thymine anion (in DMSO)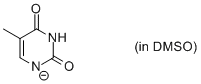  |
DMSO | N Param.: 17.63 sN Param.: 0.62 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
2-Bn super-dmap (in MeCN)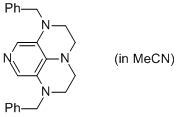  |
MeCN | N Param.: 17.69 sN Param.: 0.57 | Org. Lett. 2011, 13, 530-533 10.1021/ol1029589 | |
methylhydrazine (in MeCN)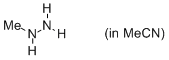  |
MeCN | N Param.: 17.73 sN Param.: 0.58 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
trimethylhydrazine (in MeCN)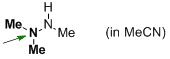  |
MeCN | N Param.: 17.75 sN Param.: 0.53 | Angew. Chem. Int. Ed. 2012, 51, 1353-1356 10.1002/anie.201107315 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in CH2Cl2)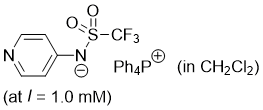  |
dichloromethane | N Param.: 17.88 sN Param.: 0.73 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
N-methylenepyrrolidin-1-amine (in CH2Cl2)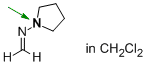  |
dichloromethane | N Param.: 17.90 sN Param.: 0.48 | Angew. Chem. Int. Ed. 2013, 52, 11900-11904 10.1002/anie.201305092 | |
dimethylamine (in MeCN)  |
MeCN | N Param.: 17.96 sN Param.: 0.63 | J. Org. Chem. 2012, 77, 8142-8155 10.1021/jo301497g | |
adenine anion (in DMSO)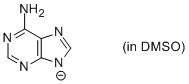  |
DMSO | N Param.: 18.00 sN Param.: 0.55 | Chem. Eur. J. 2012, 18, 127-137 10.1002/chem.201102411 | |
proline (anionic, in water)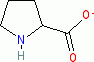  |
water | N Param.: 18.08 sN Param.: 0.50 | Org. Biomol. Chem. 2007, 5, 3814-3820 10.1039/b713778h | |
piperidine (in water)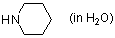  |
water | N Param.: 18.13 sN Param.: 0.44 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
perhydroazepine (in water)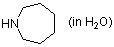  |
water | N Param.: 18.29 sN Param.: 0.46 | J. Org. Chem. 2007, 72, 3679-3688 10.1021/jo062586z | |
(S)-1-(pyrrolidin-2-ylmethyl)pyrrolidine  |
MeCN | N Param.: 18.33 sN Param.: 0.64 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
pyrrolidine (inMeCN)  |
MeCN | N Param.: 18.58 sN Param.: 0.61 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 |
