Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
p-nitrophenolate (in MeCN)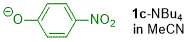  |
MeCN | N Param.: 15.14 sN Param.: 0.82 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-nitrophenolate (in DMF)  |
DMF | N Param.: 15.05 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-nitrophenolate (in DMSO)  |
DMSO | N Param.: 15.48 sN Param.: 0.71 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-nitrophenolate (in MeCN)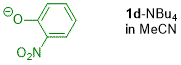  |
MeCN | N Param.: 15.83 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-nitrophenolate (in DMF)  |
DMF | N Param.: 16.65 sN Param.: 0.70 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-nitrophenolate (in DMSO)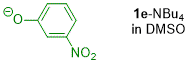  |
DMSO | N Param.: 21.29 sN Param.: 0.54 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-nitrophenolate (in MeCN)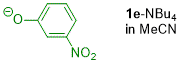  |
MeCN | N Param.: 25.51 sN Param.: 0.43 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-nitrophenolate (in DMF)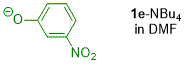  |
DMF | N Param.: 22.41 sN Param.: 0.54 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-cyanophenolate (in DMSO)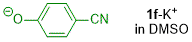  |
DMSO | N Param.: 18.78 sN Param.: 0.57 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-cyanophenolate (in MeCN)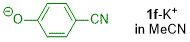  |
MeCN | N Param.: 19.58 sN Param.: 0.59 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-cyanophenolate (in DMF)  |
DMF | N Param.: 17.85 sN Param.: 0.71 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-(trifluoromethyl)phenolate (in DMSO)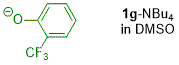  |
DMSO | N Param.: 18.47 sN Param.: 0.61 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-(trifluoromethyl)phenolate (in MeCN)  |
MeCN | N Param.: 18.39 sN Param.: 0.69 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-(trifluoromethyl)phenolate (in DMF)  |
DMF | N Param.: 20.37 sN Param.: 0.58 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(trifluoromethyl)phenolate (in DMSO)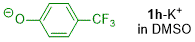  |
DMSO | N Param.: 18.98 sN Param.: 0.63 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(trifluoromethyl)phenolate (in MeCN)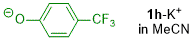  |
MeCN | N Param.: 21.69 sN Param.: 0.53 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(trifluoromethyl)phenolate (in DMF)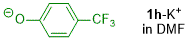  |
DMF | N Param.: 22.05 sN Param.: 0.53 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in DMSO)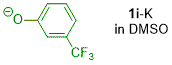  |
DMSO | N Param.: 22.62 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in MeCN)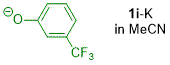  |
MeCN | N Param.: 23.20 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-(trifluoromethyl)phenolate (in DMF)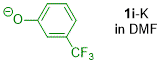  |
DMF | N Param.: 23.40 sN Param.: 0.51 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-chlorophenolate (in DMSO)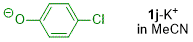  |
DMSO | N Param.: 20.34 sN Param.: 0.64 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-chlorophenolate (in MeCN)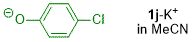  |
MeCN | N Param.: 18.32 sN Param.: 0.82 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-chlorophenolate (in DMF)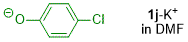  |
DMF | N Param.: 19.67 sN Param.: 0.72 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in DMSO)  |
DMSO | N Param.: 19.86 sN Param.: 0.71 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in MeCN)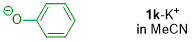  |
MeCN | N Param.: 18.53 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in DMF)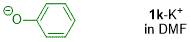  |
DMF | N Param.: 18.86 sN Param.: 0.89 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMSO)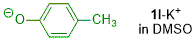  |
DMSO | N Param.: 19.08 sN Param.: 0.87 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMF)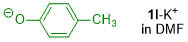  |
DMF | N Param.: 19.65 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in DMSO)  |
DMSO | N Param.: 19.67 sN Param.: 0.79 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in MeCN)  |
MeCN | N Param.: 21.70 sN Param.: 0.58 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in DMF)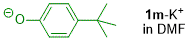  |
DMF | N Param.: 19.90 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMSO)  |
DMSO | N Param.: 20.09 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in MeCN)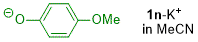  |
MeCN | N Param.: 20.62 sN Param.: 0.69 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMF)  |
DMF | N Param.: 19.90 sN Param.: 0.82 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-methoxyphenolate (in MeCN)  |
MeCN | N Param.: 18.81 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o,o'-di-tert-butylphenolate (in DMSO)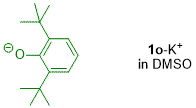  |
DMSO | N Param.: 21.02 sN Param.: 0.59 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in H2O)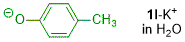  |
water | N Param.: 14.30 sN Param.: 0.55 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
4-nitrothiophenolate (in DMSO)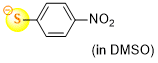  |
DMSO | N Param.: 18.92 sN Param.: 0.87 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
3,5-bis(trifluoromethyl)thiophenolate (in DMSO)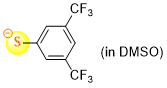  |
DMSO | N Param.: 19.71 sN Param.: 0.86 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
4-(trifluoromethyl)thiophenolate (in DMSO)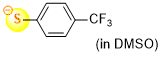  |
DMSO | N Param.: 21.30 sN Param.: 0.86 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
3-(trifluoromethyl)thiophenolate (in DMSO)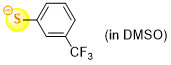  |
DMSO | N Param.: 21.75 sN Param.: 0.86 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
3-chlorothiophenolate (in DMSO)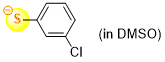  |
DMSO | N Param.: 22.50 sN Param.: 0.78 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
4-bromothiophenolate (in DMSO)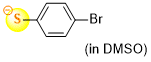  |
DMSO | N Param.: 22.80 sN Param.: 0.78 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
thiophenolate (in DMSO)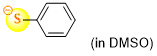  |
DMSO | N Param.: 23.36 sN Param.: 0.74 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
1,3-dimethyl-2-methyleneimidazolidine (in THF)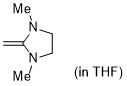  |
THF | N Param.: 18.11 sN Param.: 0.68 | J. Org. Chem. 2021, 86, 2974-2985 10.1021/acs.joc.0c02838 | |
1,3-dimethyl-2-methylenehexahydropyrimidine (in THF)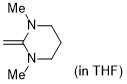  |
THF | N Param.: 18.68 sN Param.: 0.63 | J. Org. Chem. 2021, 86, 2974-2985 10.1021/acs.joc.0c02838 | |
1,3-dimesityl-2-methylene-2,3-dihydro-1H-imidazole (in THF)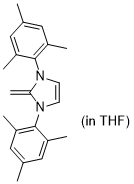  |
THF | N Param.: 17.80 sN Param.: 0.79 | J. Org. Chem. 2021, 86, 2974-2985 10.1021/acs.joc.0c02838 | |
1,3-dimethyl-2-methylene-2,3-dihydro-1H-benzo[d]imidazole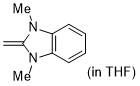  |
THF | N Param.: 19.84 sN Param.: 0.58 | J. Org. Chem. 2021, 86, 2974-2985 10.1021/acs.joc.0c02838 | |
4-methylthiophenolate (in DMSO)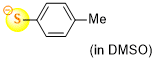  |
DMSO | N Param.: 24.35 sN Param.: 0.69 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 | |
naphthalene-2-thiolate (in DMSO)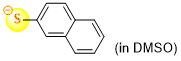  |
DMSO | N Param.: 22.55 sN Param.: 0.83 | J. Org. Chem. 2021, 86, 5965-5972 10.1021/acs.joc.1c00025 |
