N-Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
2-phenyl-4,5-dihydro-1H-imidazole (in CH2Cl2)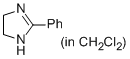  |
dichloromethane | N Param.: 12.31 sN Param.: 0.77 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
1-(2-methyl-4,5-dihydro-1H-imidazol-1-yl)ethanone (in CH2Cl2)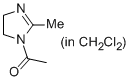  |
dichloromethane | N Param.: 10.03 sN Param.: 0.75 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
1-benzyl-2-phenyl-4,5-dihydro-1H-imidazole (in CH2Cl2)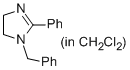  |
dichloromethane | N Param.: 13.11 sN Param.: 0.83 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-4,5-dihydrothiazole (in CH2Cl2)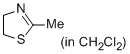  |
dichloromethane | N Param.: 10.20 sN Param.: 0.71 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-4,5-dihydro-1,3-oxazole (in CH2Cl2)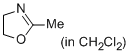  |
dichloromethane | N Param.: 9.81 sN Param.: 0.77 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-4,5-dihydro-3H-pyrrole (in CH2Cl2)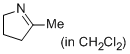  |
dichloromethane | N Param.: 13.12 sN Param.: 0.69 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-1,4,5,6-tetrahydropyrimidine (in CH2Cl2)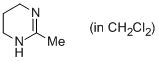  |
dichloromethane | N Param.: 15.21 sN Param.: 0.69 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-1,4,5,6-tetrahydropyrimidine (in MeCN)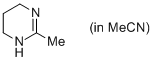  |
MeCN | N Param.: 14.43 sN Param.: 0.79 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-methyl-1,4,5,6-tetrahydropyrimidine (in DMSO)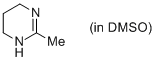  |
DMSO | N Param.: 14.58 sN Param.: 0.79 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
2-phenyl-1,4,5,6-tetrahydropyrimidine (in CH2Cl2)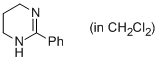  |
dichloromethane | N Param.: 14.62 sN Param.: 0.72 | Eur. J. Org. Chem. 2013, , 3369-3377 10.1002/ejoc.201300213 | |
N-methyl-1-phenylmethanimine (in CH2Cl2)  |
dichloromethane | N Param.: 8.60 sN Param.: 0.77 | Z. Naturforsch. B 2013, 68b, 693-699 10.5560/ZNB.2013-3085 | |
N-phenylpropan-2-imine (in CH2Cl2)  |
dichloromethane | N Param.: 9.53 sN Param.: 0.85 | Z. Naturforsch. B 2013, 68b, 693-699 10.5560/ZNB.2013-3085 | |
N-(phenylmethyl)propan-2-imine (in CH2Cl2)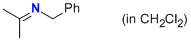  |
dichloromethane | N Param.: 11.13 sN Param.: 0.73 | Z. Naturforsch. B 2013, 68b, 693-699 10.5560/ZNB.2013-3085 | |
N-phenylcyclohexanimine (in CH2Cl2)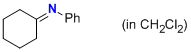  |
dichloromethane | N Param.: 8.80 sN Param.: 1.00 | Z. Naturforsch. B 2013, 68b, 693-699 10.5560/ZNB.2013-3085 | |
1,1-dimethyl-2-methylenehydrazine (in CH2Cl2)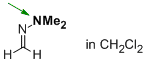  |
dichloromethane | N Param.: 19.31 sN Param.: 0.46 | Angew. Chem. Int. Ed. 2013, 52, 11900-11904 10.1002/anie.201305092 | |
N-methylenepyrrolidin-1-amine (in CH2Cl2)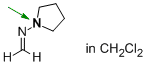  |
dichloromethane | N Param.: 17.90 sN Param.: 0.48 | Angew. Chem. Int. Ed. 2013, 52, 11900-11904 10.1002/anie.201305092 | |
4-(2-(fluorodiphenylmethyl)pyrrolidin-1-yl)pyridine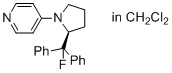  |
dichloromethane | N Param.: 14.57 sN Param.: 0.75 | Eur. J. Org. Chem. 2014, , 1202-1211 10.1002/ejoc.201301730 | |
2-phenylethylamine (in water)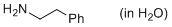  |
water | N Param.: 13.40 sN Param.: 0.57 | ChemPlusChem 2015, 80, 1673-1679 10.1002/cplu.201500246 | |
nicotine (in CH2Cl2)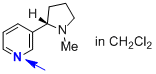  |
dichloromethane | N Param.: 10.40 sN Param.: 1.04 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
nicotine (in MeCN)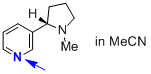  |
MeCN | N Param.: 11.60 sN Param.: 0.81 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
3-methylpyridine (in CH2Cl2)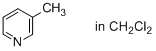  |
dichloromethane | N Param.: 10.90 sN Param.: 0.93 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
3-methylpyridine (in MeCN)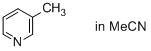  |
MeCN | N Param.: 11.50 sN Param.: 0.80 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
1-methyl-2-phenylpyrrolidine (in CH2Cl2)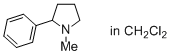  |
dichloromethane | N Param.: 16.80 sN Param.: 0.49 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
1-methyl-2-phenylpyrrolidine (in MeCN)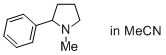  |
MeCN | N Param.: 15.70 sN Param.: 0.54 | J. Phys. Org. Chem. 2016, 29, 759-767 10.1002/poc.3580 | |
4-(morpholino)pyridine (in CH2Cl2)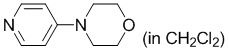  |
dichloromethane | N Param.: 14.59 sN Param.: 0.69 | Eur. J. Org. Chem. 2016, , 4050-4058 10.1002/ejoc.201600572 | |
prolinate (in MeCN)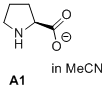  |
MeCN | N Param.: 19.95 sN Param.: 0.68 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-methylpyrrolidine (in MeCN)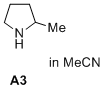  |
MeCN | N Param.: 16.78 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
pyrrolidine (inMeCN)  |
MeCN | N Param.: 18.58 sN Param.: 0.61 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(R)-2-isopropylpyrrolidine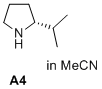  |
MeCN | N Param.: 16.44 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2,2-dimethylpyrrolidine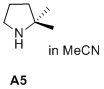  |
MeCN | N Param.: 13.96 sN Param.: 0.76 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-benzylpyrrolidine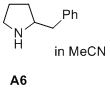  |
MeCN | N Param.: 17.43 sN Param.: 0.66 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-benzhydrylpyrrolidine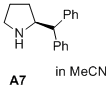  |
MeCN | N Param.: 16.61 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-pyrrolidin-2-ylmethanamine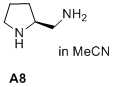  |
MeCN | N Param.: 17.24 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-N,N-dimethyl-1-(pyrrolidin-2-yl)methanamine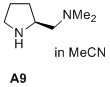  |
MeCN | N Param.: 17.41 sN Param.: 0.68 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(pyrrolidin-2-ylmethyl)pyrrolidine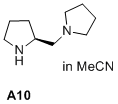  |
MeCN | N Param.: 18.33 sN Param.: 0.64 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(azidomethyl)pyrrolidine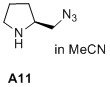  |
MeCN | N Param.: 15.43 sN Param.: 0.73 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-pyrrolidin-2-ylmethanol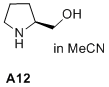  |
MeCN | N Param.: 16.74 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(methoxymethyl)pyrrolidine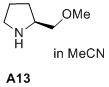  |
MeCN | N Param.: 16.50 sN Param.: 0.71 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-(trifluoromethyl)pyrrolidine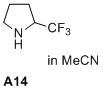  |
MeCN | N Param.: 11.34 sN Param.: 0.73 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
methyl L-prolinate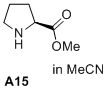  |
MeCN | N Param.: 14.75 sN Param.: 0.82 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-N,N-dimethylpyrrolidine-2-carboxamide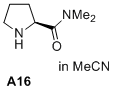  |
MeCN | N Param.: 17.61 sN Param.: 0.67 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-N-propylpyrrolidine-2-carboxamide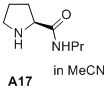  |
MeCN | N Param.: 15.20 sN Param.: 0.73 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(pyrrolidin-2-ylmethyl)isoindoline-1,3-dione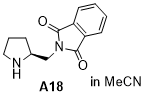  |
MeCN | N Param.: 15.90 sN Param.: 0.77 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-4-phenyl-1-(pyrrolidin-2-ylmethyl)-1H-1,2,3-triazole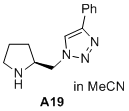  |
MeCN | N Param.: 15.32 sN Param.: 0.72 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(pyrrolidin-2-ylmethyl)-1H-imidazole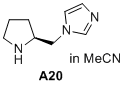  |
MeCN | N Param.: 15.55 sN Param.: 0.69 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-3-butyl-1-(pyrrolidin-2-ylmethyl)-1H-imidazol-3-ium trifluoromethanesulfonate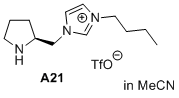  |
MeCN | N Param.: 13.57 sN Param.: 0.53 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(3,5-bis(trifluoromethyl)phenyl)-3-(pyrrolidin-2-ylmethyl)thiourea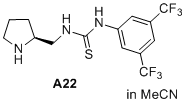  |
MeCN | N Param.: 14.97 sN Param.: 0.69 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-1-(3,5-bis(trifluoromethyl)phenyl)-3-(pyrrolidin-2-ylmethyl)urea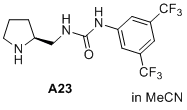  |
MeCN | N Param.: 17.50 sN Param.: 0.64 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
2-tritylpyrrolidine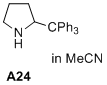  |
MeCN | N Param.: 9.16 sN Param.: 1.39 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 | |
(S)-2-(diphenyl((trimethylsilyl)oxy)methyl)pyrrolidine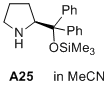  |
MeCN | N Param.: 12.03 sN Param.: 0.98 | J. Am. Chem. Soc. 2020, 142, 1526-1547 10.1021/jacs.9b11877 |
