Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
phenolate (in DMSO)  |
DMSO | N Param.: 19.86 sN Param.: 0.71 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in MeCN)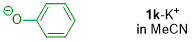  |
MeCN | N Param.: 18.53 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
phenolate (in DMF)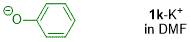  |
DMF | N Param.: 18.86 sN Param.: 0.89 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMSO)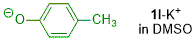  |
DMSO | N Param.: 19.08 sN Param.: 0.87 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in DMF)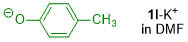  |
DMF | N Param.: 19.65 sN Param.: 0.85 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in DMSO)  |
DMSO | N Param.: 19.67 sN Param.: 0.79 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in MeCN)  |
MeCN | N Param.: 21.70 sN Param.: 0.58 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-(tert-butyl)phenolate (in DMF)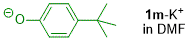  |
DMF | N Param.: 19.90 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMSO)  |
DMSO | N Param.: 20.09 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in MeCN)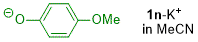  |
MeCN | N Param.: 20.62 sN Param.: 0.69 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methoxyphenolate (in DMF)  |
DMF | N Param.: 19.90 sN Param.: 0.82 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
m-methoxyphenolate (in MeCN)  |
MeCN | N Param.: 18.81 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o,o'-di-tert-butylphenolate (in DMSO)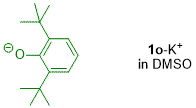  |
DMSO | N Param.: 21.02 sN Param.: 0.59 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
p-methylphenolate (in H2O)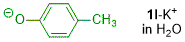  |
water | N Param.: 14.30 sN Param.: 0.55 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
glutathione GSH(NH3+/S-)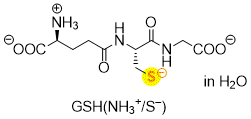  |
water | N Param.: 20.97 sN Param.: 0.56 | Angew. Chem. Int. Ed. 2019, 58, 17704-17708 10.1002/anie.201909803 | |
(E)-1-styrylpyrrolidine (in MeCN)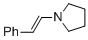  |
MeCN | N Param.: 13.87 sN Param.: 0.76 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-1-(2-(4-methoxyphenyl)-1-phenylvinyl)pyrrolidine (in MeCN)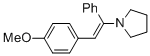  |
MeCN | N Param.: 11.99 sN Param.: 0.84 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-1-(1,2-diphenylvinyl)pyrrolidine (in MeCN)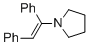  |
MeCN | N Param.: 11.66 sN Param.: 0.82 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-4-(2-phenyl-2-(pyrrolidin-1-yl)vinyl)benzonitrile (in MeCN)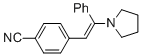  |
MeCN | N Param.: 10.63 sN Param.: 0.84 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-1-(2-(4-nitrophenyl)-1-phenylvinyl)pyrrolidine (in MeCN)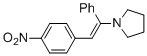  |
MeCN | N Param.: 10.42 sN Param.: 0.82 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-1-(1,2-diphenylvinyl)piperidine (in MeCN)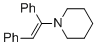  |
MeCN | N Param.: 9.94 sN Param.: 0.86 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-4-(1,2-diphenylvinyl)morpholine (in MeCN)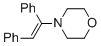  |
MeCN | N Param.: 8.78 sN Param.: 0.83 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-beta-(N-piperidino)styrene (in MeCN)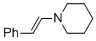  |
MeCN | N Param.: 13.84 sN Param.: 0.73 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
(E)-4-styrylmorpholine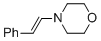  |
MeCN | N Param.: 11.66 sN Param.: 0.83 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
alpha-(N-piperidino)styrene (in MeCN)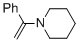  |
MeCN | N Param.: 11.60 sN Param.: 0.80 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
alpha-(N-morpholino)styrene (in MeCN)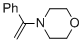  |
MeCN | N Param.: 10.30 sN Param.: 0.80 | Chem. Eur. J. 2018, 24, 5901-5910 10.1002/chem.201705962 | |
hypochlorite (in water)  |
water | N Param.: 14.50 sN Param.: 0.58 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
hypobromite (in water)  |
water | N Param.: 16.69 sN Param.: 0.46 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
bromite (in water)  |
water | N Param.: 12.75 sN Param.: 0.59 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
peroxomonosulfate (in water)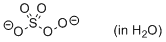  |
water | N Param.: 14.41 sN Param.: 0.60 | Org. Lett. 2018, 20, 2816-2820 10.1021/acs.orglett.[...] | |
(p-nitrophenyl)diazomethane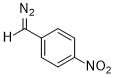  |
dichloromethane | N Param.: 7.17 sN Param.: 0.83 | J. Am. Chem. Soc. 2018, 140, 16758-16772 10.1021/jacs.8b09995 | |
(p-cyanophenyl)diazomethane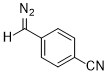  |
dichloromethane | N Param.: 7.66 sN Param.: 0.80 | J. Am. Chem. Soc. 2018, 140, 16758-16772 10.1021/jacs.8b09995 | |
(p-bromophenyl)diazomethane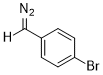  |
dichloromethane | N Param.: 8.87 sN Param.: 0.82 | J. Am. Chem. Soc. 2018, 140, 16758-16772 10.1021/jacs.8b09995 | |
beta-piperidinium-peroxypropionate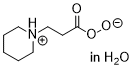  |
water | N Param.: 13.94 sN Param.: 0.62 | Eur. J. Org. Chem. 2018, , 6010-6017 10.1002/ejoc.201801158 | |
peroxy-beta-alanate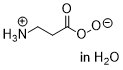  |
water | N Param.: 14.07 sN Param.: 0.56 | Eur. J. Org. Chem. 2018, , 6010-6017 10.1002/ejoc.201801158 | |
peroxy-GABA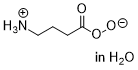  |
water | N Param.: 14.33 sN Param.: 0.57 | Eur. J. Org. Chem. 2018, , 6010-6017 10.1002/ejoc.201801158 | |
1-ethoxy-3-(4-nitrophenyl)-1,3-dioxopropan-2-ide (in DMSO)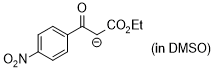  |
DMSO | N Param.: 16.26 sN Param.: 0.83 | J. Am. Chem. Soc. 2018, 140, 11474-11486 10.1021/jacs.8b07147 | |
methanol (in MeCN)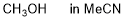  |
MeCN | N Param.: 6.86 sN Param.: 0.73 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
ethanol (in MeCN)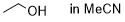  |
MeCN | N Param.: 7.13 sN Param.: 0.71 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
isopropanol (in MeCN)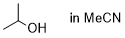  |
MeCN | N Param.: 6.82 sN Param.: 0.70 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
tert-butanol (in MeCN)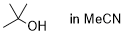  |
MeCN | N Param.: 5.35 sN Param.: 0.72 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
water (in MeCN)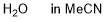  |
MeCN | N Param.: 5.79 sN Param.: 0.72 | Bull. Chem. Soc. Jpn. 2018, 91, 523-530 10.1246/bcsj.20170360 | |
chloro(phenylsulfonyl)methanide (in DMSO)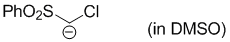  |
DMSO | N Param.: 28.27 sN Param.: 0.42 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
bromo(phenylsulfonyl)methanide (in DMSO)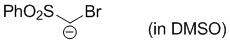  |
DMSO | N Param.: 23.90 sN Param.: 0.62 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro((4-cyanophenyl)sulfonyl)methanide (in DMSO)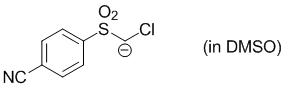  |
DMSO | N Param.: 25.59 sN Param.: 0.51 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro((4-nitrophenyl)sulfonyl)methanide (in DMSO)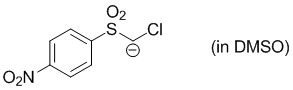  |
DMSO | N Param.: 24.88 sN Param.: 0.49 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
chloro((4-chlorophenyl)sulfonyl)methanide (in DMSO)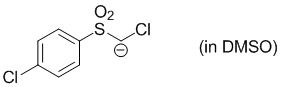  |
DMSO | N Param.: 26.90 sN Param.: 0.45 | J. Org. Chem. 2017, 82, 2011-2017 10.1021/acs.joc.6b02844 | |
anion of ethyl 2-cyano-2-phenylacetate (in DMSO)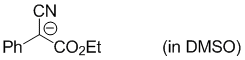  |
DMSO | N Param.: 15.85 sN Param.: 1.04 | Eur. J. Org. Chem. 2017, , 1196-1202 10.1002/ejoc.201601513 | |
anion of 2-phenylmalononitrile (in DMSO)  |
DMSO | N Param.: 15.58 sN Param.: 1.00 | Eur. J. Org. Chem. 2017, , 1196-1202 10.1002/ejoc.201601513 | |
anion of 2-phenyl-2-(phenylsulfonyl)acetonitrile (in DMSO)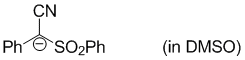  |
DMSO | N Param.: 15.97 sN Param.: 0.72 | Eur. J. Org. Chem. 2017, , 1196-1202 10.1002/ejoc.201601513 |
