Electrophiles
1 | 2 | 3 | 4 | 5 | 6 | 7 | 8
« Back Forward » Found 377 molecules, displaying page 3 of 8 Results per page
10|50|100|all
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
pop(Ph)CH+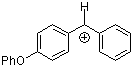  |
E Param.: 2.90 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y | ||
tol(Ph)CH+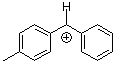  |
E Param.: 4.43 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(pfp)2CH+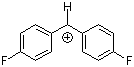  |
E Param.: 5.01 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
fc(Ph)CH+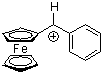  |
E Param.: -2.64 | J. Am. Chem. Soc. 2001, 123, 9500-9512 10.1021/ja010890y | ||
tritylium ion (Ph3C+)  |
E Param.: 0.51 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-CF3)2-tritylium ion  |
E Param.: 2.28 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(3-Cl)3-tritylium ion  |
E Param.: 1.99 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-CF3)-tritylium ion  |
E Param.: 1.33 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(3-Cl)-tritylium ion  |
E Param.: 1.06 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(3-CF3)-tritylium ion  |
E Param.: 1.18 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-Me)-tritylium ion  |
E Param.: -0.13 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-Me)2-tritylium ion  |
E Param.: -0.70 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-Me)3-tritylium ion  |
E Param.: -1.21 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-MeO,3-OMe)-tritylium ion  |
E Param.: -1.62 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-OMe,3-Me)-tritylium ion  |
E Param.: -1.84 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-MeO,4-Me)-tritylium ion  |
E Param.: -2.13 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-MeO)2-tritylium ion  |
E Param.: -3.04 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-MeO)3-tritylium ion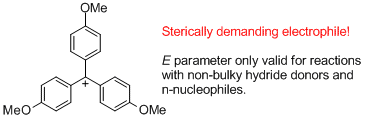  |
E Param.: -4.35 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-NMe2)-tritylium ion  |
E Param.: -7.93 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-NMe2)2-tritylium ion (= Malachite green)  |
E Param.: -10.29 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(4-NMe2)3-tritylium ion (= Crystal violet)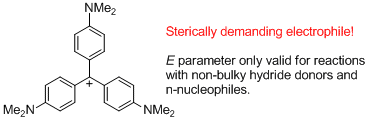  |
E Param.: -11.26 | J. Am. Chem. Soc. 2003, 125, 286-295 10.1021/ja021010y | ||
(E)-3-(4-nitro-phenyl)-1-phenyl-prop-2-en-1-one (in DMSO)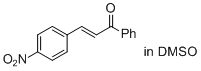  |
DMSO | E Param.: -17.33 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-3-(4-cyano-phenyl)-1-phenyl-prop-2-en-1-one (in DMSO)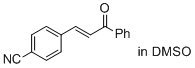  |
DMSO | E Param.: -17.64 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-1,3-diphenylprop-2-en-1-one (in DMSO)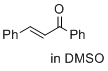  |
DMSO | E Param.: -19.39 | J. Am. Chem. Soc. 2017, 139, 13318-13329 10.1021/jacs.7b05106 | |
(E)-4-(4-nitrophenyl)but-3-en-2-one (in DSMO)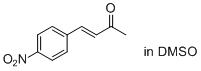  |
DMSO | E Param.: -19.36 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-4-methyl-1-(4-nitrophenyl)pent-1-en-3-one (in DMSO)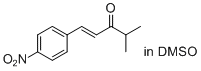  |
DMSO | E Param.: -19.17 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-4,4-dimethyl-1-(4-nitrophenyl)pent-1-en-3-one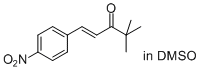  |
DMSO | E Param.: -19.15 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
3-chloro-benzaldehyde (in DMSO)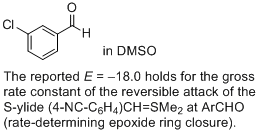  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
3-fluoro-benzaldehyde (in DMSO)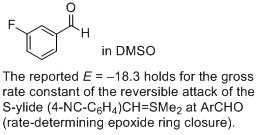  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
3-methoxy-benzaldehyde (in DMSO)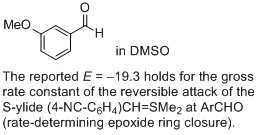  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
4-fluoro-benzaldehyde (in DMSO)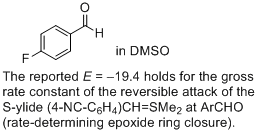  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
benzaldehyde (in DMSO)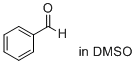  |
DMSO | E Param.: -12.90 | J. Am. Chem. Soc. 2018, 140, 5500-5515 10.1021/jacs.8b01657 | |
hexanal (in DMSO)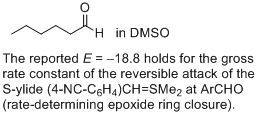  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
butanal (in DMSO)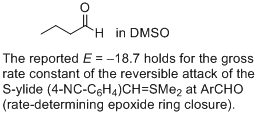  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
cinnamaldehyde (in DMSO)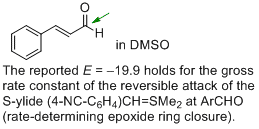  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
p-nitro-cinnamaldehyde (in DMSO)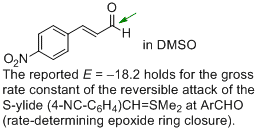  |
DMSO | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | ||
(E)-N-benzylidene-4-methylbenzenesulfonamide (in DMSO)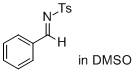  |
DMSO | E Param.: -11.50 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-N-(4-methoxybenzylidene)-4-methylbenzenesulfonamide (in DMSO)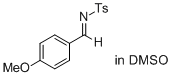  |
DMSO | E Param.: -13.05 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-N-(4-(dimethylamino)benzylidene)-4-methylbenzenesulfonamide (in DMSO)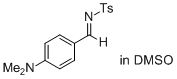  |
DMSO | E Param.: -15.09 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-tert-butyl benzylidenecarbamate (in DMSO)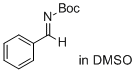  |
DMSO | E Param.: -14.22 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(E)-N-benzylidene-P,P-diphenylphosphinic amide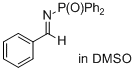  |
DMSO | E Param.: -15.89 | J. Am. Chem. Soc. 2011, 133, 8240-8251 10.1021/ja200820m | |
(fur)(ani)CH+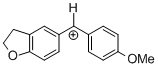  |
E Param.: -0.81 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(mfp)PhCH+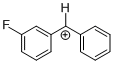  |
E Param.: 6.23 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(tfm)PhCH+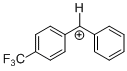  |
E Param.: 6.70 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(dfp)PhCH+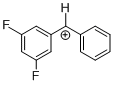  |
E Param.: 6.74 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(mfp)2CH+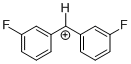  |
E Param.: 6.87 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(dfp)(mfp)CH+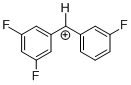  |
E Param.: 7.52 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(tfm)2CH+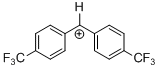  |
E Param.: 7.96 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(dfp)2CH+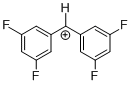  |
E Param.: 8.02 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b | ||
(Ftol)2CH+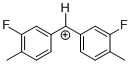  |
E Param.: 5.24 | J. Am. Chem. Soc. 2012, 134, 13902-13911 10.1021/ja306522b |
