Nucleophiles
| Name | Solvent |
Reactivity Parameters |
Classification |
Reference (title or year) |
|---|---|---|---|---|
(F)2-HBTM (in CH2Cl2)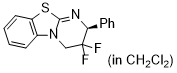  |
dichloromethane | N Param.: 10.83 sN Param.: 0.80 | ARKIVOC 2024, (4), 202312093 10.24820/ark.5550190[...] | |
2-methylpyridine (in CH2Cl2)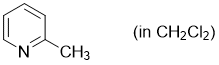  |
dichloromethane | N Param.: 10.52 sN Param.: 0.78 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
2,6-dimethylpyridine (in CH2Cl2)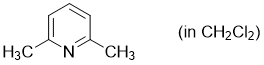  |
dichloromethane | N Param.: 9.87 sN Param.: 0.68 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
2,4,6-trimethylpyridine (in CH2Cl2)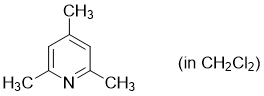  |
dichloromethane | N Param.: 9.34 sN Param.: 0.71 | Synthesis 2017, 49, 3495-3504 10.1055/s-0036-1590504 | |
methyl diazoacetate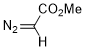  |
dichloromethane | N Param.: 4.68 sN Param.: 0.94 | J. Am. Chem. Soc. 2023, 145, 7416-7434 10.1021/jacs.2c13872 | |
dimethyl diazomalonate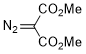  |
dichloromethane | N Param.: -1.24 sN Param.: 0.81 | J. Am. Chem. Soc. 2023, 145, 7416-7434 10.1021/jacs.2c13872 | |
pyridin-4-yl((trifluoromethyl)sulfonyl)amide (Ph4P+) (in CH2Cl2)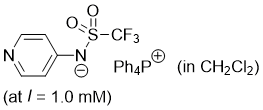  |
dichloromethane | N Param.: 17.88 sN Param.: 0.73 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
((4-methoxyphenyl)sulfonyl)(pyridin-4-yl)amide (Ph4P+) (in CH2Cl2)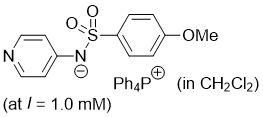  |
dichloromethane | N Param.: 19.63 sN Param.: 0.65 | J. Am. Chem. Soc. 2025, 147, 5043-5050 10.1021/jacs.4c14825 | |
O-DHPB (in CH2Cl2)  |
dichloromethane | N Param.: 13.31 sN Param.: 0.69 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
O-HyperBTM (in CH2Cl2)  |
dichloromethane | N Param.: 13.01 sN Param.: 0.73 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Te-BTM (in CH2Cl2)  |
dichloromethane | N Param.: 14.62 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-DHPB (in CH2Cl2)  |
dichloromethane | N Param.: 15.16 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-tetramisole (Se-TM) (in CH2Cl2)  |
dichloromethane | N Param.: 15.26 sN Param.: 0.60 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-BTM (in CH2Cl2)  |
dichloromethane | N Param.: 14.27 sN Param.: 0.63 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HBTM (in CH2Cl2)  |
dichloromethane | N Param.: 14.42 sN Param.: 0.67 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Se-HyperBTM (in DCM)  |
dichloromethane | N Param.: 16.11 sN Param.: 0.58 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
Te-DHPB (in CH2Cl2)  |
dichloromethane | N Param.: 16.63 sN Param.: 0.65 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
tetramisole (TM) (in CH2Cl2)  |
dichloromethane | N Param.: 13.86 sN Param.: 0.68 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
BTM (in CH2Cl2)  |
dichloromethane | N Param.: 13.06 sN Param.: 0.73 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
7-F-BTM (in CH2Cl2)  |
dichloromethane | N Param.: 13.46 sN Param.: 0.66 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
7-MeO-BTM (in CH2Cl2)  |
dichloromethane | N Param.: 13.15 sN Param.: 0.75 | Angew. Chem. Int. Ed. 2025, 64, e202514865 10.1002/anie.202514865 | |
(1-azidovinyl)benzene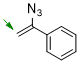  |
dichloromethane | N Param.: 4.57 sN Param.: 0.94 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
1-(1-azidovinyl)-4-methylbenzene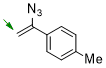  |
dichloromethane | N Param.: 5.47 sN Param.: 0.87 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
1-(1-azidovinyl)-4-methoxybenzene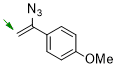  |
dichloromethane | N Param.: 6.08 sN Param.: 0.99 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
4-(1-azidovinyl)-1,1'-biphenyl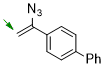  |
dichloromethane | N Param.: 5.17 sN Param.: 0.86 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
1-(1-azidovinyl)-4-(chloromethyl)benzene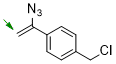  |
dichloromethane | N Param.: 4.13 sN Param.: 0.96 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
1-(1-azidovinyl)-4-(trifluoromethyl)benzene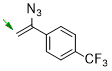  |
dichloromethane | N Param.: 3.10 sN Param.: 0.77 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
2-(1-azidovinyl)naphthalene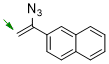  |
dichloromethane | N Param.: 5.39 sN Param.: 0.89 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
1-(1-azidovinyl)naphthalene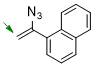  |
dichloromethane | N Param.: 3.17 sN Param.: 0.88 | Chem. Commun. 2026, 62, 5041-5045 10.1039/d6cc00190d | |
DHPB (in CH2Cl2)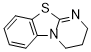  |
dichloromethane | N Param.: 13.86 sN Param.: 0.78 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
HBTM (in CH2Cl2)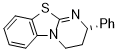  |
dichloromethane | N Param.: 13.45 sN Param.: 0.72 | J. Org. Chem. 2011, 76, 5104-5112 10.1021/jo200803x | |
Te-HyperBTM (in DCM)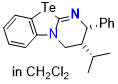  |
dichloromethane | N Param.: 16.63 sN Param.: 0.63 | ChemistryEurope 2026, 4, e202500443 10.1002/ceur.202500443 | |
3,4-dimethoxy-2-(methoxymethyl)-3,4-dihydro-2H-pyran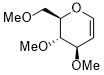  |
dichloromethane | N Param.: 1.53 sN Param.: 1.38 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
2-(acetoxymethyl)-3,4-dihydro-2H-pyran-3,4-diyl diacetate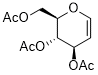  |
dichloromethane | N Param.: 1.72 sN Param.: 1.24 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-((benzyloxy)methyl)-3,4-dihydro-2H-pyran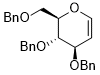  |
dichloromethane | N Param.: 1.89 sN Param.: 1.27 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
2-((TBDMSO-methyl)-3,4-bis(TBDMSO)-3,4-dihydro-2H-pyran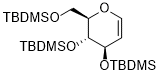  |
dichloromethane | N Param.: 1.97 sN Param.: 1.20 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-((benzyloxy)methyl)-6-methyl-3,4-dihydro-2H-pyran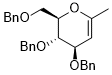  |
dichloromethane | N Param.: 4.98 sN Param.: 0.65 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-((benzyloxy)methyl)-3,4-dihydro-2H-pyran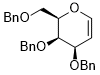  |
dichloromethane | N Param.: 1.75 sN Param.: 1.37 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-((benzyloxy)methyl)-6-methylenetetrahydro-2H-pyran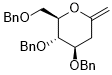  |
dichloromethane | N Param.: 6.19 sN Param.: 0.79 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
8-(benzyloxy)-2,2-di-tert-butyl-4,4a,8,8a-tetrahydropyrano[3,2-d][1,3,2]dioxasiline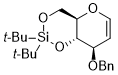  |
dichloromethane | N Param.: 1.85 sN Param.: 1.28 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4,5-tris(benzyloxy)-2-((benzyloxy)methyl)-6-methylenetetrahydro-2H-pyran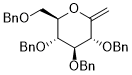  |
dichloromethane | N Param.: 4.01 sN Param.: 0.91 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-((benzyloxy)methyl)-5-methylenetetrahydrofuran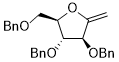  |
dichloromethane | N Param.: 5.50 sN Param.: 0.76 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
4-((benzyloxy)methyl)-2,2-dimethyl-6-methylenetetrahydrofuro[3,4-d][1,3]dioxole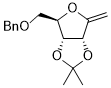  |
dichloromethane | N Param.: 5.35 sN Param.: 0.88 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
8-(benzyloxy)-2-phenyl-4,4a,8,8a-tetrahydropyrano[3,2-d][1,3]dioxine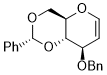  |
dichloromethane | N Param.: 1.94 sN Param.: 1.24 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
3,4-bis(benzyloxy)-2-methyl-3,4-dihydro-2H-pyran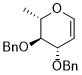  |
dichloromethane | N Param.: 2.04 sN Param.: 1.20 | Angew. Chem. Int. Ed. 2026, EarlyView, e24329 10.1002/anie.202524329 | |
1,3-bis(trimethylsilyl)propane  |
dichloromethane/MeCN mix | N Param.: -5.30 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
(4,4-dimethylpentyl)trimethylstannane  |
dichloromethane/MeCN mix | N Param.: -3.90 sN Param.: 1.10 | Chem. Eur. J. 2013, 19, 249-263 10.1002/chem.201202839 | |
4-(dimethylamino)pyridine (in DMF)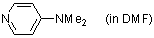  |
DMF | N Param.: 14.90 sN Param.: 0.67 | Chem. Eur. J. 2007, 13, 336-345 10.1002/chem.200600941 | |
p-nitrophenolate (in DMF)  |
DMF | N Param.: 15.05 sN Param.: 0.80 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 | |
o-nitrophenolate (in DMF)  |
DMF | N Param.: 16.65 sN Param.: 0.70 | J. Org. Chem. 2019, 84, 8837-8858 10.1021/acs.joc.9b01485 |
